- Synthesis Spotlight
- Posts
- Simple Skeletal Editing
Simple Skeletal Editing
💡 Lightning as Powerful as 2,400 Tons of TNT

Monday 23rd March – Sunday 29th March 2026 | Volume 3, Issue 12 |


Skeletal Editing by Iron-Catalyzed Carbene Insertion of Trichloromethanes
B. M. DeMuynck,† E. E. Hyland,† H. Kim,† V. L. Menches,† A. M. M. Vernier, Z. Wang, D. E. Olson,* M. D. Levin* & D. A. Nagib*
Chem 2026, Online Now (DOI: 10.1016/j.chempr.2026.102968)
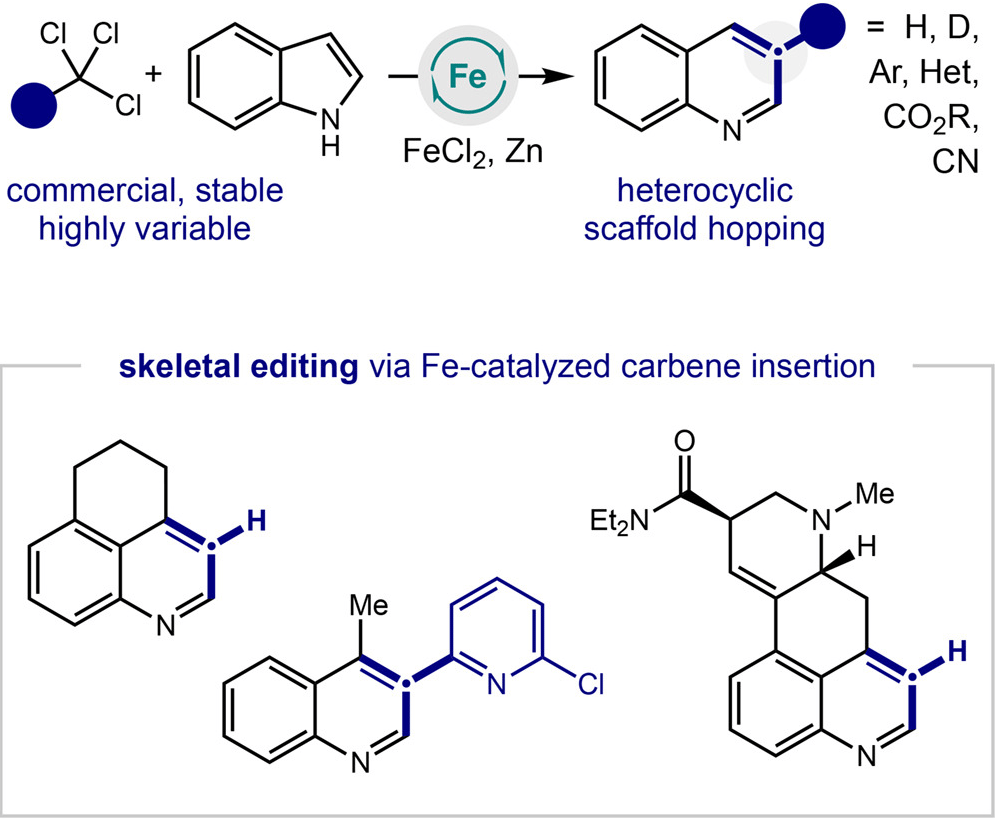
The authors report a practical C-insertion strategy that uses stable, commercially abundant trichloromethanes as chlorocarbene precursors in combination with simple FeCl2 catalysis to directly convert indoles into diverse quinolines. This operationally simple method enables rare C–H and C–D insertion, providing access to valuable medicinal chemistry analogs and other pharmacophores. The approach is applicable to complex molecules, including the one-step synthesis of a quinoline analog of lysergic acid diethylamide, which shows reduced potency and efficacy at 5-HT2A receptors—highlighting potential for therapeutic applications with diminished hallucinogenic effects.

Late-Stage Synthesis of C4-Fluorinated and C4-Trifluoromethylaminated Pyridines
L. Chen, C. Zu, F. Zhang, Y. Ding,* Y.-X. Luan,* L. Chen* & P. Tang*
Nat. Synth. 2026 (DOI: 10.1038/s44160-026-01035-2)
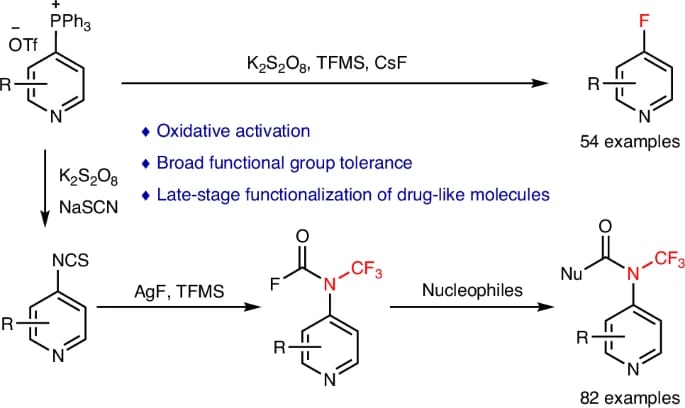
The authors report an oxidative activation strategy that enables the synthesis of C4-fluorinated and C4-trifluoromethylaminated pyridines via oxidation of pyridyl phosphonium salts from common precursors. This approach overcomes longstanding challenges associated with weak fluoride nucleophilicity, low C4 electrophilicity, and inefficient NCF3 installation, while exhibiting broad functional group tolerance and applicability to bioactive molecules. The resulting analogues display enhanced bioactivity, highlighting the method’s potential in medicinal chemistry.
Fluoroform Upcycling to Trifluorovinylamine as a C2 reagent to Prepare Difluoromethylated Molecules
P. Ivashkin,† J. Decaens,† I. Marchand, M. Hedouin, H. Oulyadi, T. Poisson* & P. Jubault*
Nat. Synth. 2026 (DOI: 10.1038/s44160-026-01001-y)
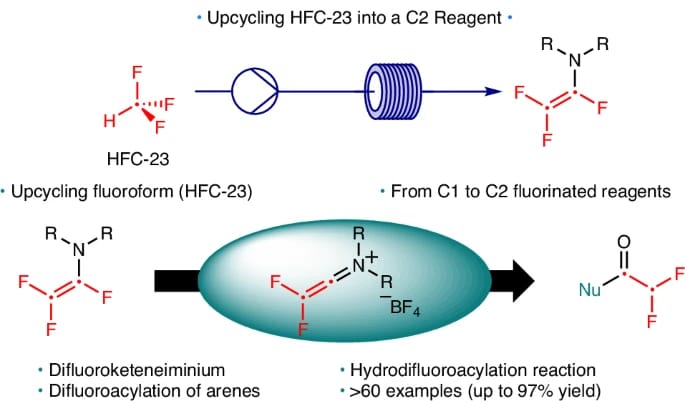
The authors report a strategy for the valorization of fluoroform (HFC-23), a highly potent greenhouse gas produced in multi-tonne quantities that has traditionally been incinerated or used as a C1 feedstock. This approach enables the synthesis of trifluorovinylamine, a novel C2 building block, from HFC-23. Trifluorovinylamine exhibits unique reactivity as a difluoroacylium surrogate, facilitating efficient formation of α,α-difluoromethyl ketones via electrophilic aromatic substitution and enabling the development of a hydrodifluoroacetylation reaction.

Photoinduced Mn Catalysis for Efficient Platform for C–Heteroatom Bond Coupling of Aryl Halides
G. Song,† J. Song,† Q. Li, X. Shi, X. Liu, D. Pan, T. Kang, J. Dong, G. Li, H. Sun, J. Fan, C. Wang & D. Xue*
Nat. Commun. 2026 (DOI: 10.1038/s41467-026-70925-y) 🔓
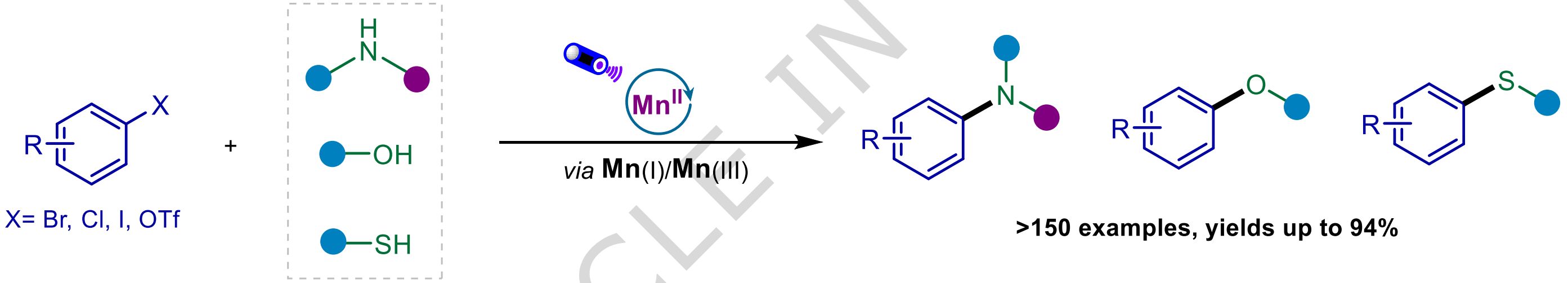
The authors report a highly efficient, photoinduced Mn(II)–bipyridine-catalyzed protocol for C–N, C–O, and C–S cross-coupling of aryl halides, including typically unreactive aryl chlorides, with a range of heteroatom nucleophiles. This method demonstrates broad substrate scope, encompassing eight classes of nitrogen nucleophiles for C–N bond formation, as well as alcohols and thiophenols for C–O and C–S coupling, respectively. Over 150 examples are reported, with yields of up to 94%.

Nickel-Catalyzed Cross-Dehydrogenative Allylation of Aldehydes
A. F. Ibrahim,† Y. H. Fujisato,† A. M. Ventura, P. M. Zimmerman* & J. Montgomery*
J. Am. Chem. Soc. 2026, ASAP (DOI: 10.1021/jacs.5c20798)
Previously: ChemRxiv (DOI: 10.26434/chemrxiv-2025-gbv0j) 🔓
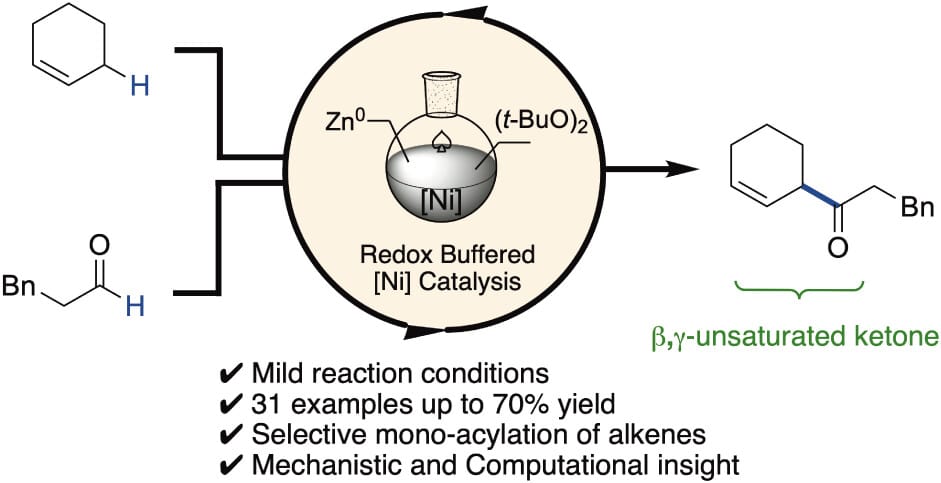
The authors describe a complementary approach to aldehyde–alkene couplings via direct nickel-catalyzed cross-dehydrogenative coupling of aldehydes and alkenes. The process employs zinc powder and di-tert-butyl peroxide to enable the synthesis of β,γ-unsaturated ketones through the catalytic union of acyl and allylic radicals generated from the respective substrates, consistent with a double hydrogen-atom-transfer mechanism. A broad array of substrate combinations is demonstrated.
Synthesis of 1,3-Dienes from Alkenes via Alkenyl Thianthrenium Salts
S. Müller, N. Klask, A. Daff & T. Ritter*
J. Am. Chem. Soc. 2026, ASAP (DOI: 10.1021/jacs.5c22101) 🔓
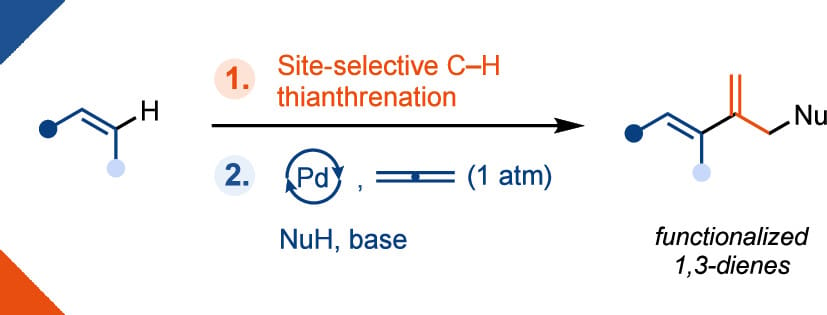
The authors report the conversion of alkenes into functionalized 1,3-dienes in two steps. First, C–H thianthrenation is used to access a wide pool of alkenyl electrophiles directly from alkenes. Second, the alkenyl thianthrenium salts undergo a Pd-catalyzed three-component coupling reaction with allene gas and non-organometallic nucleophiles to afford 1,3-dienes. This method provides a robust and modular platform for accessing structurally complex 1,3-dienes that would be challenging to access otherwise.
Synergistic Transition Metal and Hydrogen Bonding Phase-Transfer Catalysis Enables Enantioconvergent Allylic Fluorination with KF
Z. Wang, C. Dooley, Z. Chen, G. Poškaitė, R. S. Paton, G. C. Lloyd-Jones* & V. Gouverneur*
J. Am. Chem. Soc. 2026, ASAP (DOI: 10.1021/jacs.6c00549) 🔓
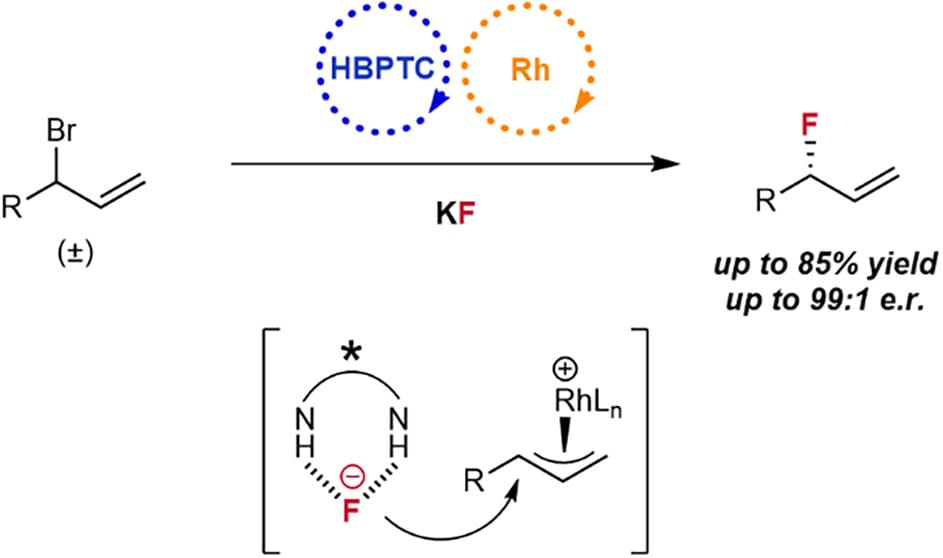
Synergistic catalysis whereby both the nucleophile and the electrophile can be simultaneously activated by two distinct catalysts can rescue otherwise unattainable chemical transformations, as well as create or improve catalytic enantioselectivity. Here, the authors report the merging of transition metal and hydrogen bonding phase-transfer catalysis to allow for allyl bromides to undergo enantioselective fluorination with KF.
Navigating the Landscape of Cycloartanyl Cations: Synthesis of Fortunefuroic Acid I, Parkeol, 25,26,27-Trinor-3α-hydroxy-17,13-friedolanosta-8,12-dien-23-one, and Spirochensilide A
M. Kizakis, M. Treger, G. Dräger, C. König & P. Heretsch*
J. Am. Chem. Soc. 2026, ASAP (DOI: 10.1021/jacs.5c22292) 🔓
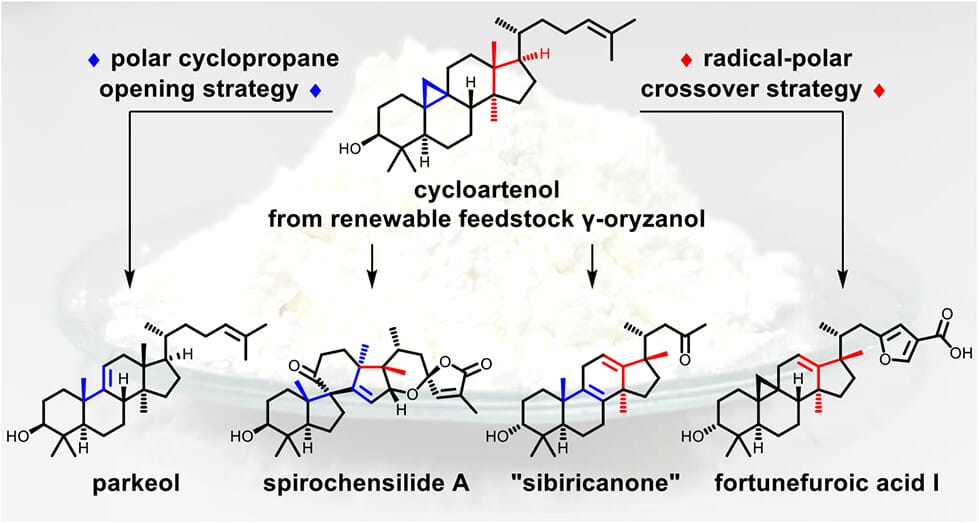
The authors report a semisynthetic approach to plant-derived triterpenoids based on a robust route to cycloartenol derivatives from the renewable feedstock γ-oryzanol. By combining radical–polar crossover and polar activation modes, distinct carbenium ions are selectively accessed and guided through rearrangements. This strategy enables a modular toolbox for stepwise skeletal reorganization, providing divergent access to parkeol-, 17,13-friedocycloartane-, and friedolanostane frameworks, and their conversion into parkeol, fortunefuroic acid I, sibiricanone, and spirochensilide A. Experimental and DFT studies further elucidate cycloartanyl cation reactivity and support a biosynthetic rationale for rearranged lanostane derivatives in plants.
Programmed Oxidation-Guided Divergent Total Syntheses of (±)-Anisatin, (±)-Neoanisatin, (±)-Majusanols A and B
J. Huang, X. Wei, Z. Chen & M. Yang*
J. Am. Chem. Soc. 2026, ASAP (DOI: 10.1021/jacs.6c02820)
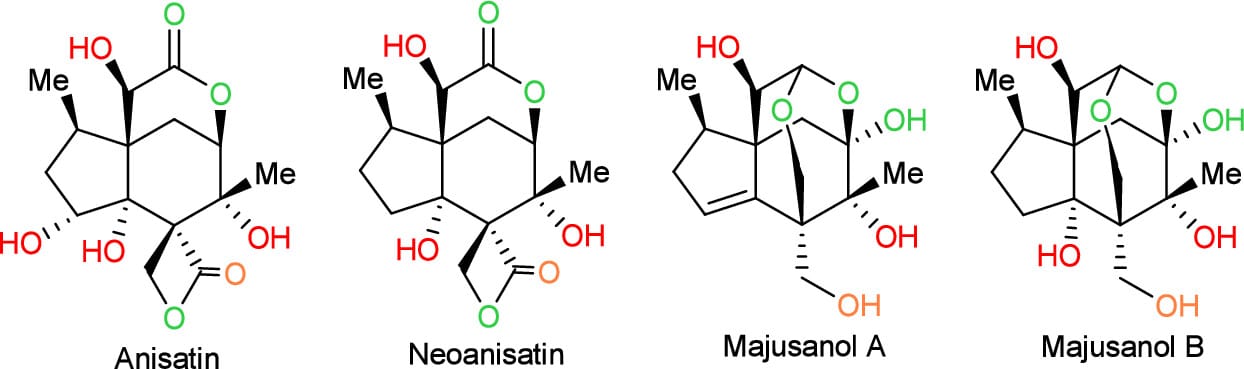
The authors report a programmed oxidation strategy that prioritizes oxygen installation according to its role in skeletal construction. This approach enables the first total syntheses of neoanisatin (20 steps), majusanol A (18 steps), and majusanol B (19 steps), and provides a significantly shorter route to anisatin (20 steps vs. prior 40- and 43-step syntheses).

Radical Retrosynthesis of Natural Products Enabled by Iron-Based Reductive Olefin Coupling
G. L. Barnes,† A. L. Rerick† & P. S. Baran*
Angew. Chem. Novit 2026, 2, e70017 (DOI: 10.1002/anov.70017) 🔓
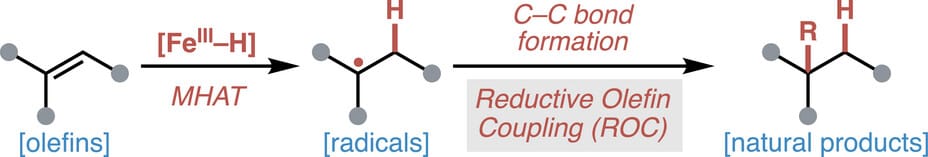
This review highlights iron-catalyzed reductive olefin coupling (ROC)—a strategy for C—C bond formation driven by metal-hydride hydrogen atom transfer (MHAT)—as a key tool in natural products synthesis. This review surveys dozens of total syntheses employing ROC-based strategies, highlighting its power to forge strained rings, quaternary centers, and stereocenters from olefins, often supplanting 2e− tactics.
Navigating Nitration Chemistry: A Practical Guide to Reagents, Mechanisms, and Selectivity
H. Lecomte, A. J. Fernandes & D. Katayev*
Angew. Chem. Int. Ed. 2026, Early View (DOI: 10.1002/anie.202526128) 🔓
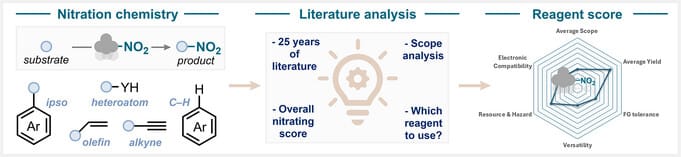
This review highlights key contributions of modern nitration chemistry, emphasizing sustainable mechanistic platforms and comparing the performance of both organic and inorganic reagents across aromatic, ipso-, olefin, alkyne, and heteroatom nitration. It provides the community with a clearer, unified perspective on current advances and facilitates the selection of nitrating reagents by establishing a performance score.

1,2-Migration of Oxamoyl-Protected Alcohols via Reversible C–H Sampling
Y. Su, D. Wang, K. Chen & Y. Xu*
ChemRxiv 2026 (DOI: 10.26434/chemrxiv.15001327/v1) 🔓
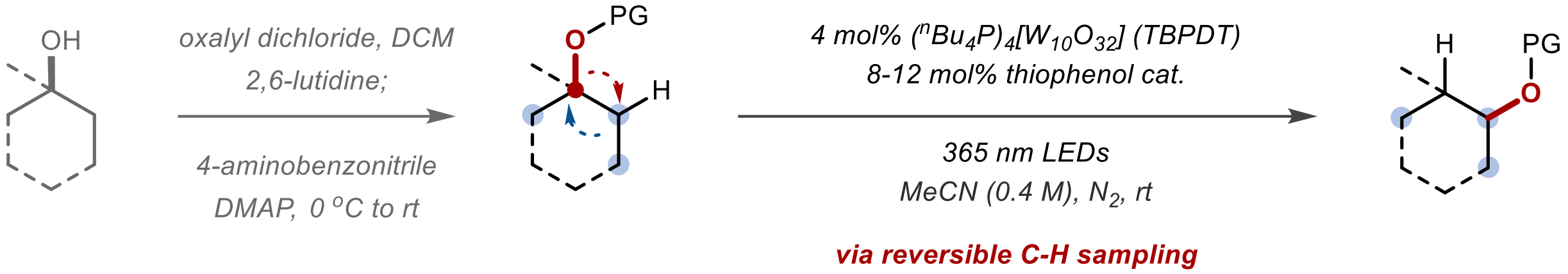
The authors report a photocatalytic 1,2-translocation of oxamoyl-protected alcohols enabled by a reversible C–H sampling strategy. Using a cooperative hydrogen atom transfer catalytic system, transient carbon radicals are generated, with β-acyloxy alkyl radicals undergoing a selective 1,2-acyloxy shift to furnish rearranged products. The oxamoyl protecting group plays a crucial role in promoting the rearrangement and enhancing β-H abstraction efficiency. This redox-neutral method exhibits broad substrate scope with predictable regio- and stereoselectivity, including late-stage modification of complex bioactive molecules.
Safe Cyanation of Alcohols Enabled by Synchronized Photocatalytic Nucleophile and Electrophile Generation
T. Banik,† I. A. Dmitriev,† M. Belil-Catalina, B. Limburg* & B. Pieber*
ChemRxiv 2026 (DOI: 10.26434/chemrxiv.15001128/v1) 🔓
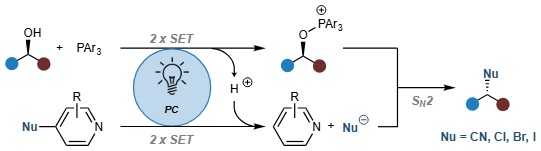
The authors present a photocatalytic manifold for the cyanation of alcohols that recreates Mitsunobu-type reactivity without relying on hazardous reagents. The reaction design is based on the photocatalytic generation of labile phosphoranyl radicals that typically undergo rapid β-scission to yield carbon-centered radicals; however, in the presence of cyanopyridines, this fragmentation pathway is outcompeted by a single-electron transfer process, leading to the formation of an oxyphosphonium cation and in situ cyanide generation that engage in an SN2 reaction. This mechanistic blueprint is further extended to Appel-type halogenations.
Denitrogenative Glycosylations of Glycosyl Sulfonohydrazides
S. Zhao,† S. Chen,† H. Ma, Y. Nian* B. Yang* & F. Zhu*
ChemRxiv 2026 (DOI: 10.26434/chemrxiv.15001154/v1) 🔓

The authors report a nickel-catalysed, stereoselective denitrogenative glycosylation of redox-neutral glycosyl sulfonohydrazides via anomeric C–N bond homolysis. Bench-stable glycosyl sulfonohydrazides are readily synthesized via an open–closed ring equilibrium without stoichiometric reagents and intrinsically generate glycosyl radicals without external redox additives. This operationally simple platform enables highly stereoselective formation of diverse C–C, C–S, C–Se and C–Te bonds with broad substrate scope and excellent functional group tolerance.
👉️ This work builds upon the use of glycosyl sulfonyl hydrazides for radical-mediated C-glycoside synthesis as reported by Baran, Aggarwal and co-workers here.
Newman-Kwart Rearrangement Enabled by Flavin Photoredox Catalysis
C. D. Glenn,† M. Sharma† & K. F. Biegasiewicz*
ChemRxiv 2026 (DOI: 10.26434/chemrxiv.15001191/v1) 🔓
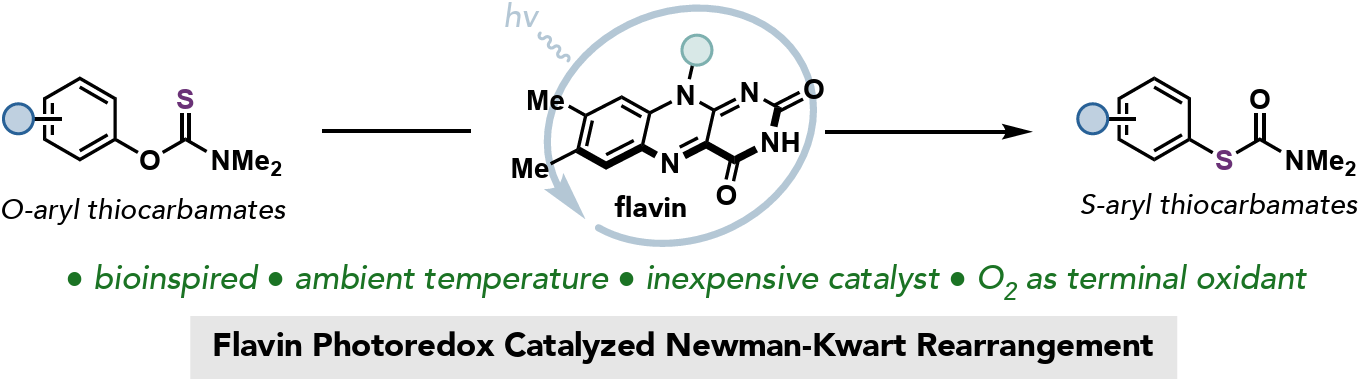
The Newman-Kwart Rearrangement (NKR) is a valuable transformation for accessing thiophenol derivatives from readily available phenols. Traditional methods for conducting the NKR are often limited by harsh reaction conditions, operational scalability, and reactions run in primarily organic solvents. Here, the authors introduce a bioinspired approach to perform the NKR using (−)-riboflavin as a cheap and effective photocatalyst. The reaction has been demonstrated on a broad range of substrates in high yield with low catalyst loadings in primarily aqueous media.
Photogenerated Oxetanes as a Gateway to Uphill Cyclopropanation
T. Köglmeier,† T. Tiefel† & O. Reiser*
ChemRxiv 2026 (DOI: 10.26434/chemrxiv.15001190/v1) 🔓
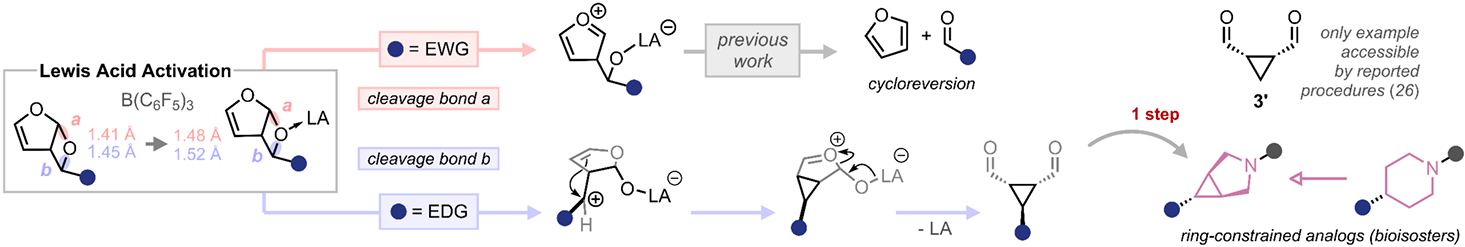
The authors report an overall endergonic, catalytic route to cyclopropanes from abundant, renewable starting materials. Furan oxetanes, readily formed by photochemical [2+2] cycloaddition of aldehydes and furan, undergo an intramolecular SN-type rearrangement under Lewis-acid catalysis, producing cyclopropanes with ideal atom economy. The strategy enables access to meso-cyclopropane dicarbaldehydes, which are versatile intermediates that streamline the preparation of otherwise challenging bioactive motifs.

10 Trillion Joules
⚡️ 10 Trillion Joules. New data from NASA’s Juno mission reveals that lightning on Jupiter is far more powerful than previously confirmed—reaching 100 to 10,000× the energy of Earth’s lightning bolts.
Using Juno’s microwave radiometer, researchers were able to peer through the planet’s dense cloud cover and directly measure radio emissions from lightning for the first time. The most extreme flashes release up to 10 trillion joules (comparable to 2,400 tons of TNT) and occur at rates of ~3 flashes per second.
Like on Earth, Jupiter’s lightning likely arises from charge separation in turbulent clouds. However, its chemistry is much more unusual: water mixes with ammonia to form slushy hail-like “mushballs”, potentially enabling far greater charge build-up and more violent discharges.
These findings matter because lightning is a key driver of complex chemistry, potentially kick-starting prebiotic reactions. The sheer energy of Jupiter’s storms suggests that lightning on other planets could play a major role in shaping atmospheric chemistry or even the origins of life.
That’s all for this issue! Have a great week and we’ll see you next Monday.

Reply