- Synthesis Spotlight
- Posts
- Shaking Up Alcohols
Shaking Up Alcohols
💡 Can a Daily Multivitamin Slow Ageing?

Monday 9th March – Sunday 15th March 2026 | Volume 3, Issue 10 |


Alcohol Group Migration by Proximity-Enhanced H Atom Abstraction
Q. Xu,† Y. Nie,† J.-J. Haaksma, R. Zhang, N. Holmberg-Douglas, F. van der Mei, P. M. Scola, C. Williams, J. A. Johnson & A. E. Wendlandt*
Nature 2026 (DOI: 10.1038/s41586-026-10347-4)
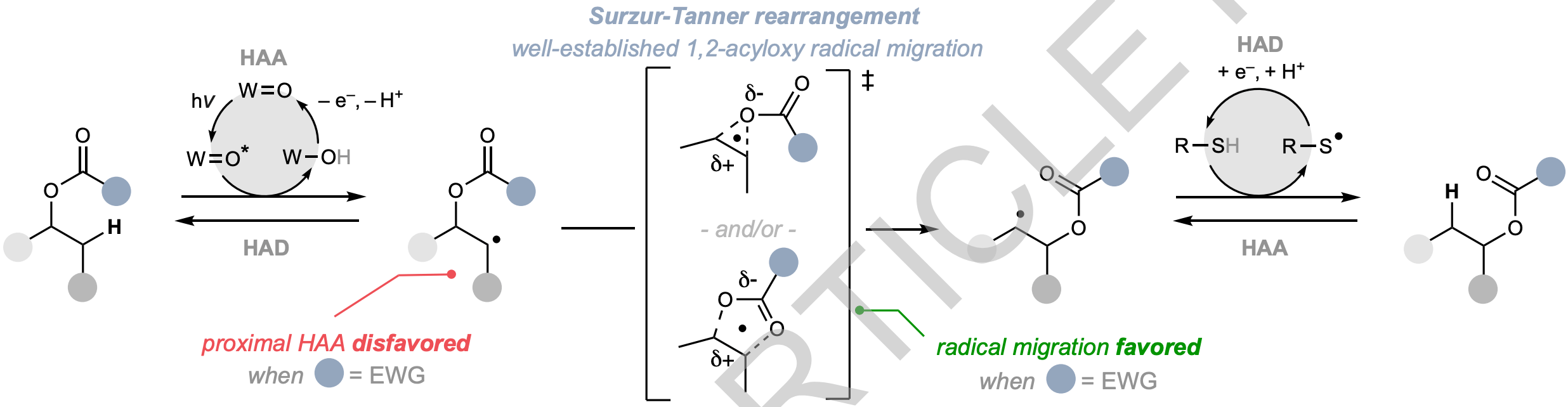
The authors report an editing method that enables the migration of common alcohol functional groups to proximal sites with predictable stereo- and regiochemical outcomes. The reaction proceeds through a 1,2-acyloxy radical migration step under reversible H atom transfer catalysis conditions promoted by excited state decatungstate polyanion. Proximity effects arising from non-covalent interactions between substrate and reagent enable efficient radical formation at polarity-mismatched positions. Application of this tool at a late synthetic stage allows for the precise re-positioning of alcohol functional groups, while integration with common alcohol group installation methods provides new synthetic strategies to access challenging oxygenation patterns.

Biocatalytic Site- and Stereoselective Carbonyl Desaturation for Late-Stage Functionalization of Cyclic Ketones
S. Cao,† Y. Zhu,† J. Lei,† R. Dai, T. Zhu* & Y. Ye*
Nat. Chem. 2026 (DOI: 10.1038/s41557-026-02086-w)
Previously: ChemRxiv (DOI: 10.26434/chemrxiv-2025-q7j3b) 🔓

The authors report the engineering of “ene”-reductases for the direct carbonyl desaturation of diverse cyclic ketones to their corresponding enones with excellent site divergence. The synthetic utility of this platform is further demonstrated through the successful late-stage dehydrogenation on terpenoids with complementary site selectivity to existing methods. In addition, the method could efficiently prepare chiral enones with a β-all carbon quaternary stereogenic centre via biocatalytic desaturative kinetic resolution.

Positional Editing of Diverse Functional Groups Through Radical 1,2-Boron Shifts
W. Zhang,† S. Mao,† M. Peng & Y. Xi*
Nat. Synth. 2026 (DOI: 10.1038/s44160-026-01018-3)
Previously: ChemRxiv (DOI: 10.26434/chemrxiv-2025-61whz) 🔓

The authors report a catalytic 1,2-transposition of a pinacol boryl group that occurs across a wide range of substrates. By harnessing the versatility of organoboron compounds as synthetic intermediates, this method enables a three-step platform for formal positional editing of a diverse set of functional groups, allowing access to corresponding positional isomers.
Anti-Friedel–Crafts Alkylation via Electron Donor–Acceptor Photoinitiation
D. M. Vahey, M. Mu, S. A. Bonke, T. Sommer, P. Vangal, C. Mallia, M. García-Melchor* & E. Reisner*
Nat. Synth. 2026 (DOI: 10.1038/s44160-026-00994-w) 🔓
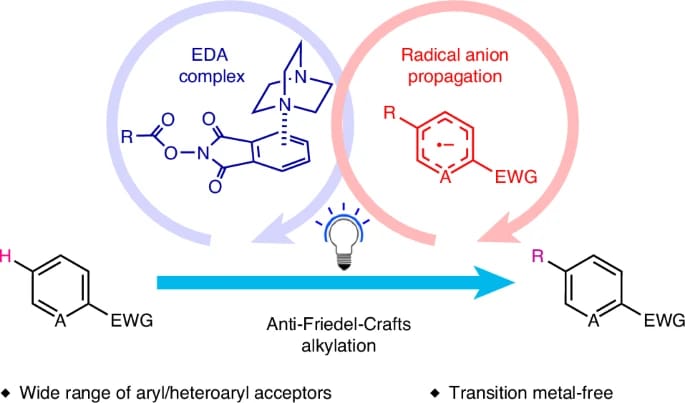
The authors introduce a selective, scalable and transition-metal-free strategy for C–H alkylation of electron-poor aromatics under mild conditions with broad functional group tolerance, enabling late-stage functionalization of pharmaceutical compounds. The mechanistic design exploits a redox-active phthalimide ester tag to form an electron donor–acceptor complex that fragments upon photoexcitation to generate a nucleophilic alkyl radical, which selectively alkylates the most electrophilic position of electron-deficient aromatics, exhibiting “anti-Friedel–Crafts” selectivity and enabling alkylation of a wide range of complex molecules.
Heteronuclear Dual-Atom Insertion into Oxetanes via Frustrated Lewis Pair Activation
Y.-Q. Zhang, S.-H. Li & M. J. Koh*
Nat. Synth. 2026 (DOI: 10.1038/s44160-026-01031-6)
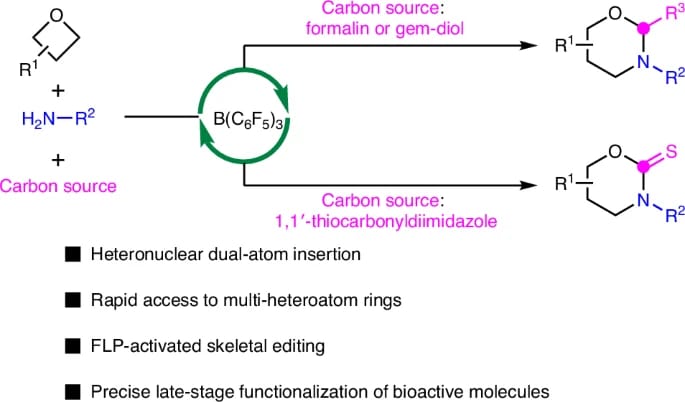
The authors report a boron-catalyzed dual-atom insertion into oxetanes using various nitrogen and carbon sources to access 1,3-oxazinanes. The method streamlines the preparation of bioactive scaffolds and is amenable to late-stage editing to generate multi-heteroatom cyclic molecules. Mechanistic studies reveal a cascade pathway in which an in situ-generated frustrated Lewis pair enables ring deconstruction and reconstruction.

Aminyl Radical-Enabled Photoredox/Nickel-Catalyzed C(sp3 )–C(sp3 ) Suzuki–Miyaura Cross-Coupling via Halogen-Atom Transfer Strategy
Y.-W. Zhang, C.-L. Ji, A. Chen, J.-X. Bi & D.-W. Gao*
J. Am. Chem. Soc. 2026, ASAP (DOI: 10.1021/jacs.6c00526)
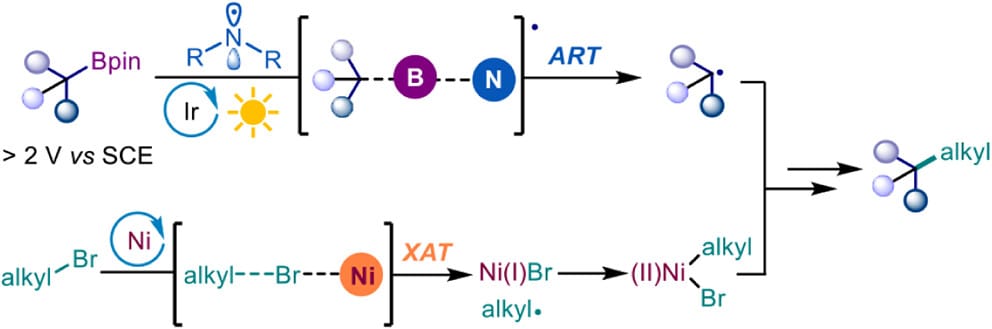
The authors utilize aminyl radicals to activate alkyl boronic esters, generating alkyl radicals under photoredox conditions; concurrently, a nickel catalyst activates alkyl electrophiles to facilitate the formation of C(sp3 )–C(sp3 ) bonds. The strategy operates under mild conditions and exhibits a broad substrate scope with excellent functional group tolerance. Furthermore, this method enables site-selective alkylation, alkenylation, alkynylation, and arylation of 1,n-bis(boronic) esters. The robustness and synthetic utility of the method are demonstrated through scale-up and continuous-flow experiments, downstream transformations, and the synthesis of a bioactive molecule.
Boron-Mediated Hydroalkylation of Unactivated Olefins: An Anti-Markovnikov Approach to Congested Carbon Centers
H. Zhang,† R. Sang,† G. Simionato,† J. L. Tyler, A. Noble* & V. K. Aggarwal*
J. Am. Chem. Soc. 2026, ASAP (DOI: 10.1021/jacs.6c01091) 🔓
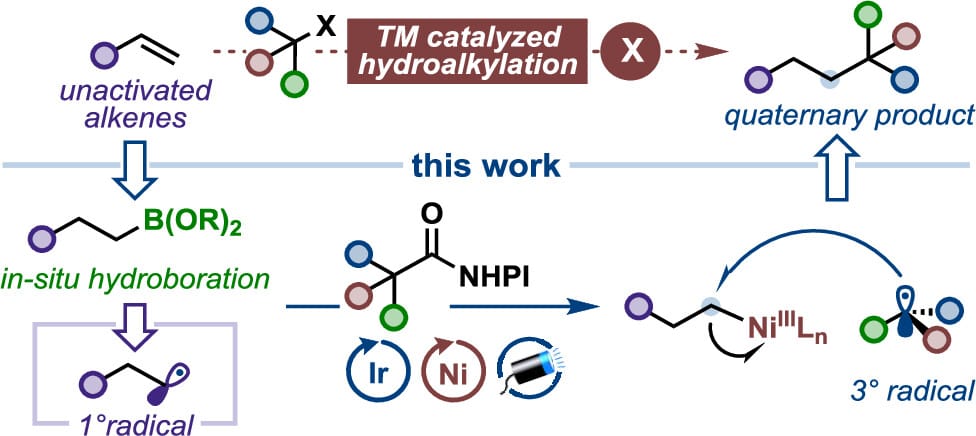
Transition metal-catalyzed hydroalkylations provide efficient routes for installing alkyl fragments onto readily available olefins. However, forming quaternary carbon centers via hydroalkylation with tertiary alkyl electrophiles remains a significant synthetic challenge due to steric congestion, which slows oxidative addition and promotes deleterious β-hydride elimination. Here, the authors report a boron-mediated coupling strategy using nickel/photoredox dual catalysis that overcomes these limitations and enables anti-Markovnikov hydroalkylation of unactivated olefins with carboxylic acid derivatives as alkylating partners.
Enantioselective Copper-Catalyzed Synthesis of Hydroxylamines via Hydrofunctionalization of Alkenes using Nitroalkanes
J. A. Law, B. K. Mai, H.-M. Hung, T. Hendon, Y. Dong, Y. Zhang, P. Liu* & S. L. Buchwald*
J. Am. Chem. Soc. 2026, ASAP (DOI: 10.1021/jacs.5c20561) 🔓
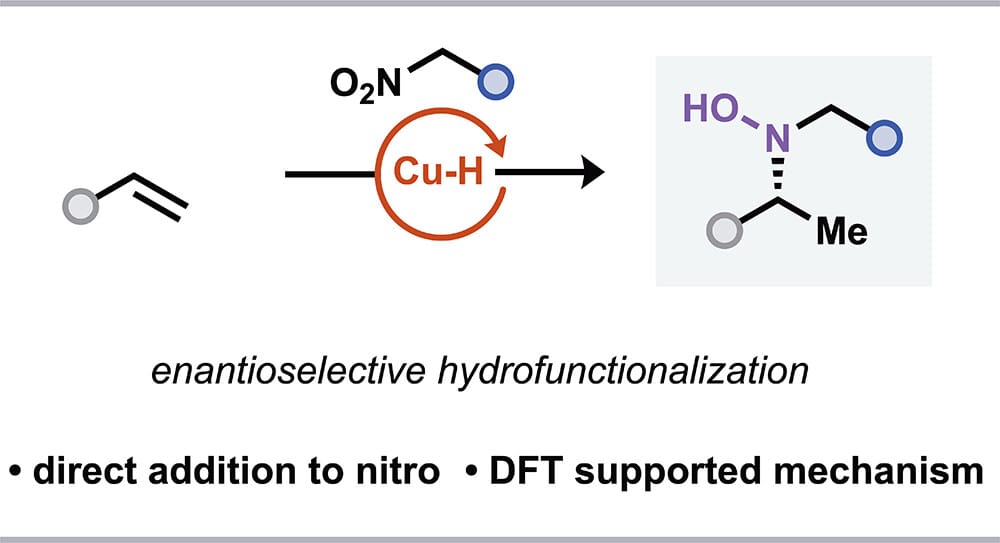
The authors report that nitroalkanes can serve as competent electrophiles in the enantioselective CuH-catalyzed hydrofunctionalization of vinyl(hetero)arenes, affording hydroxylamines in good yields and with high enantioselectivity. Control experiments and DFT calculations indicate that the nitro group acts as the active electrophile and that addition of the stereoenriched alkylcopper intermediate proceeds through a six-membered cyclic transition state involving dearomatization of the vinyl arene.
Ortho–Meta and Para–Meta Isomerization of Phenols
S. Malik, S. N. Ullal, J. D. Hart, M. Sodoor, P. A. Hume & P. S. Grant*
J. Am. Chem. Soc. 2026, ASAP (DOI: 10.1021/jacs.6c00742)
Previously: ChemRxiv (DOI: 10.26434/chemrxiv-2025-f1853) 🔓

The authors present an approach to the para–meta and ortho–meta isomerization of phenols, combining oxidative dearomatization with photochemical rearrangement and reductive aromatization. This method allows the transposition of neighboring carbon atoms within the aromatic core and enables access to challenging meta-functionalized phenols from readily available para- and ortho-isomers.
Ring Expansion of Benzofurans by Single-Silicon Insertion
Q. Yan, H. Song, H. Lu* & H. Wei*
J. Am. Chem. Soc. 2026, ASAP (DOI: 10.1021/jacs.6c00274)

The authors report a Ni-catalyzed single-silicon atom insertion reaction for the transformation of readily available benzofurans into their corresponding high-value oxasilacycles. The reaction involves the C–H bond silylation of benzofurans and 1,2-oxygen migration. The resulting oxasilacycles serve as versatile synthons for generating various compounds and provide access to different cyclic skeletons such as oxaborins, coumarins, benzooxepines, and polycyclic oxasilaalkanes.
Convergent Total Synthesis of Caribbean Ciguatoxin C-CTX1 and Its C3-Epimer
M. Sasaki,* A. Umehara, K. Sugahara, M. Satake & T. Tsumuraya
J. Am. Chem. Soc. 2026, ASAP (DOI: 10.1021/jacs.6c01247) 🔓

Caribbean ciguatoxin C-CTX1 is a potent ladder-shaped polycyclic ether toxin responsible for ciguatera fish poisoning and has recently been detected beyond the Caribbean Sea. Owing to its structural complexity and scarcity from natural sources, its structure and biological properties have not been fully validated by chemical means. Here, the authors report the first total synthesis of C-CTX1 and its C3-epimer through a convergent fragment-coupling strategy. The work provides structural confirmation, milligram-scale access to both epimers, and reveals significant differences in relative neurotoxicity.
Catalytic Asymmetric Total Synthesis of Grisemycin, a Thioangucycline Polyketide
K. Stevenson, D. P. Moran & T. J. Maimone*
J. Am. Chem. Soc. 2026, ASAP (DOI: 10.1021/jacs.6c00348)
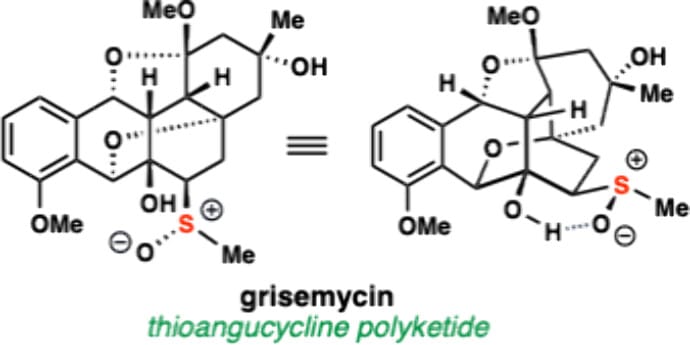
Angucycline natural products have been extensively studied for decades yet members possessing sulfur atoms (i.e. thioangucyclines) remain rare. In 2016, Zhang and co-workers isolated the structurally unprecedented thioangucycline grisemycin, from the marine actinomycete Streptomyces griseus. Grisemycin features a distinctive angucycline topology and exotic methylsulfoxide functional group of unknown biosynthetic origins. Here, the authors report the first total synthesis of this polyketide in enantioenriched form and suggest that grisemycin’s caged skeleton can assemble from a genetically encoded angucycline Michael acceptor in the presence of methanethiol and an oxidant without enzymatic assistance.
Divergent Syntheses of the Euphorbia Diterpenoids Pepluanol A and Pepluacetal
D. Qiu,† Z. Zhao,† L. Liu, Z. Xie, H.-W. Lin, L.-Z. Huang, F. Yang* & Y. Sun*
J. Am. Chem. Soc. 2026, ASAP (DOI: 10.1021/jacs.6c01271)
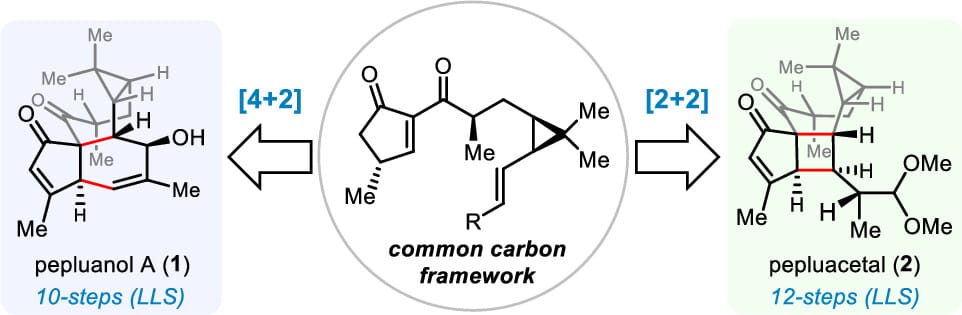
Pepluanol A and pepluacetal belong to a structurally unique class of pepluanol-type Euphorbia diterpenoids that exhibit immunosuppressive activity by targeting Kv1.3 channels. Here, the authors report a divergent strategy enabling efficient and concise total syntheses of both natural products from a common advanced intermediate. The synthesis of pepluanol A features an intramolecular Diels–Alder reaction for framework construction, whereas the synthesis of pepluacetal relies on a photoinduced intramolecular [2+2] cycloaddition as the key step. Further biological evaluation showed that these diterpenoids and their derivatives possess antipulmonary fibrosis activity.

Dual-Ligand System for Mild Decarbonylative Suzuki–Miyaura Cross-Coupling of Aroyl Chlorides
M. H. Sak, Y. Jiang, E. N. Jacobsen* & R. Y. Liu*
ACS Catal. 2026, ASAP (DOI: 10.1021/acscatal.5c08167) 🔓
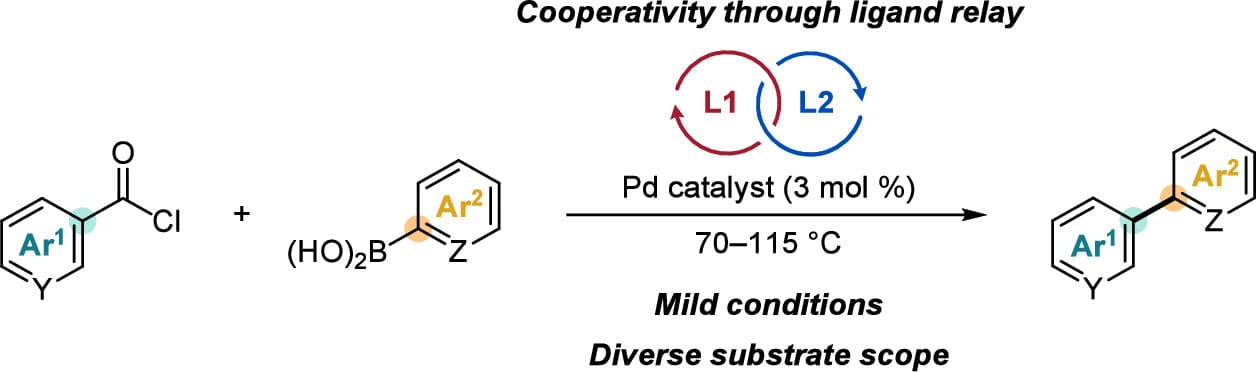
Cooperativity between a pair of phosphine ligands enables general Pd-catalyzed decarbonylative Suzuki–Miyaura cross-couplings between (hetero)aroyl chlorides and (hetero)arylboronic acids under mild conditions. Experimental and computational studies support a ligand-relay mechanism in which each phosphine preferentially promotes different elementary steps, enhancing the yield and selectivity relative to using either ligand alone.

Decoding Reactions in Process Chemistry: A Quarter-Century Analysis of Trends and Transformations in OPR&D
K. A. Iyer, S. Roy, U. T. Balande, A. Maind, H. Chen, S. P. Kolis,* Q. Yang* & Q. A. Zhou*
Org. Process Res. Dev. 2026, ASAP (DOI: 10.1021/acs.oprd.6c00001) 🔓
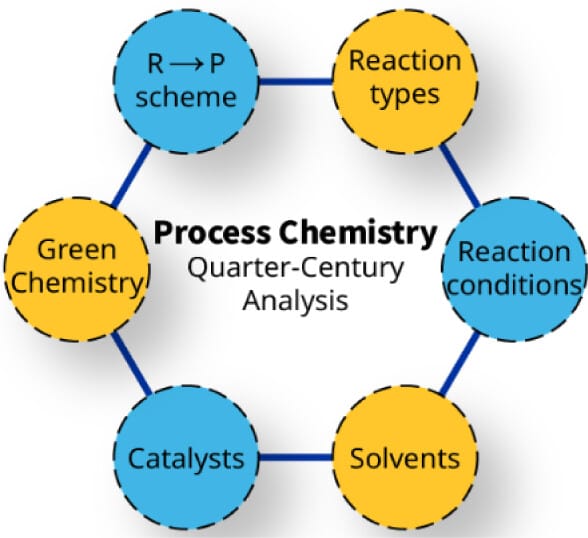
This study presents a comprehensive analysis of synthetic chemistry practices in process development through a systematic examination of Organic Process Research & Development (OPR&D) publications from 2000–2025 indexed in the CAS Content Collection. Over 4,800 articles encompassing more than 34,000 reactions were analyzed to identify trends in synthetic methods, reagents, solvents, and reaction conditions used in scalable processes. While cross-coupling reactions dominate the literature, acylation and alkylation are the most frequently performed transformations in practice. Palladium-based catalysts predominate in metal-catalyzed processes and EDC is the most common coupling reagent for amide bond formation. Flow chemistry has also emerged as an increasingly prominent enabling technology.

Ring-Opening Decarbonylative C(sp3 )–C(sp3 ) Cross-Electrophile Coupling of Cyclic Imides with Unactivated Alkyl Chlorides
N. J. Lentelink, P. M. F. Pânzar, N. A. V. Rowlinson & B. Morandi*
Chem. Sci. 2026, Accepted (DOI: 10.1039/D6SC00815A) 🔓
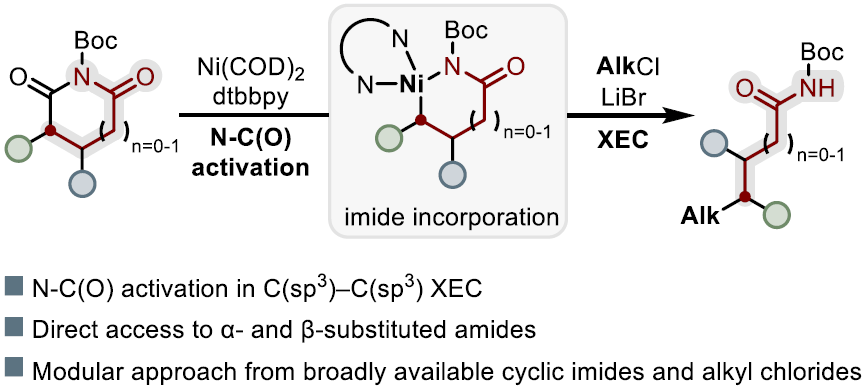
The authors report a nickel-mediated decarbonylative cross-electrophile coupling of N-Boc succinimides and glutarimides with unactivated alkyl chlorides. The transformation proceeds via selective endocyclic N–C(O) activation, providing a new entry into C(sp3 )–C(sp3 ) cross-electrophile coupling and a modular platform for rapidly constructing molecular complexity through incorporation of the ring-opened imide scaffold. Broad functional group tolerance affords structurally diverse α- and β-substituted amides, exemplified by the synthesis of densely substituted carbocyclic amides and novel capsaicin precursors.

Smallest Bicycles in Medicinal Chemistry: Where Are We Now?
V. Kubyshkin* & P. K. Mykhailiuk*
Chem. Rev. 2026, ASAP (DOI: 10.1021/acs.chemrev.5c00779)

This Review highlights the use of the smallest bicyclic and spirocyclic ring systems as saturated scaffolds for constructing bioactive molecules. The authors discuss their molecular strain and potential as saturated benzene bioisosteres in terms of geometry and volume, supported by structural data from small-molecule and protein crystallography. Synthetic strategies, the commercial availability of functionalized building blocks and examples from drug discovery programs are also surveyed. At least eight molecules incorporating these compact frameworks have advanced to clinical trials, with one (vanzacaftor) recently receiving FDA approval. The analysis also reveals a striking imbalance in scaffold adoption: while bicyclo[1.1.1]pentane and spiro[3.3]heptane have become routine tools in medicinal chemistry, structures such as ladderanes and housanes remain largely unexplored.

Winding Back the Clock
💊 Winding Back the Clock. A large randomised clinical trial has found that taking a daily multivitamin may slow biological ageing in older adults. Researchers analysing blood samples from 958 participants in the COcoa Supplement and Multivitamin Outcomes Study (COSMOS) trial found that two years of multivitamin supplementation slowed the progression of certain epigenetic ageing markers by roughly four months compared with placebo.
The analysis focused on DNA-methylation–based epigenetic clocks, molecular biomarkers that estimate biological age by tracking age-related chemical changes to DNA, and found that daily multivitamin use significantly slowed two clocks associated with mortality risk and age-related disease.
The effect was modest overall but strongest in individuals whose biological age exceeded their chronological age, suggesting those with accelerated ageing may benefit most. Cocoa flavanol supplementation, which was also tested in the trial, showed no measurable impact on the epigenetic ageing measures. Although the findings provide some of the most encouraging evidence yet that a simple supplement can influence ageing biomarkers, researchers warn that it remains unclear whether these molecular changes translate into meaningful improvements in long-term health or lifespan.
That’s all for this issue! Have a great week and we’ll see you next Monday.

Reply