Monday 6th April – Sunday 12th April 2026
Volume 3, Issue 14


Ambiphilic Cross-Coupling with Aryl-Bismuth Reagents
B. Roh, B. A. Williams & J. Cornella*
Nature 2026 (DOI: 10.1038/s41586-026-10486-8)
Previously: ChemRxiv (DOI: 10.26434/chemrxiv-2025-74b8f) 🔓

Cross-coupling reactions traditionally form Ar–Ar bonds between aryl nucleophiles and electrophiles under transition-metal catalysis. Their high selectivity relies on a clear mechanistic distinction between these partners, which undergo fundamentally different catalytic steps. Here, the authors report ambiphilic aryl-bismuth reagents capable of functioning as either nucleophiles or electrophiles in transition-metal-catalysed cross-couplings, thereby challenging this dichotomy. This behaviour arises from their ability to participate in both oxidative addition and transmetalation with transition metal complexes, as supported by stoichiometric and mechanistic studies.

Modular Enantioselective Photocatalysts from Privileged PyBox Scaffolds
R. M. Kelch, L. Hämmerling, E. Zysman-Colman* & T. P. Yoon*
Science 2026, 392, 188–193 (DOI: 10.1126/science.aeb5832)
Previously: ChemRxiv (DOI: 10.26434/chemrxiv-2025-x7lc6) 🔓

Modern organic synthesis relies on chiral catalysts to control stereochemistry, with certain “privileged” catalyst families exhibiting broad applicability across diverse transformations. However, highly enantioselective photocatalyst structures remain comparatively underdeveloped. Here, the authors report a family of enantioselective photocatalysts derived from pyridine bis(oxazoline) complexes modified with electron-donating carbazole units. These ligands are readily synthesized from chiral pool materials, enabling modular variation, while their donor–acceptor charge-transfer properties can be rationally tuned to optimize photocatalytic activity. The generality of this platform is demonstrated across multiple asymmetric transformations, including both photoredox and excited-state processes.

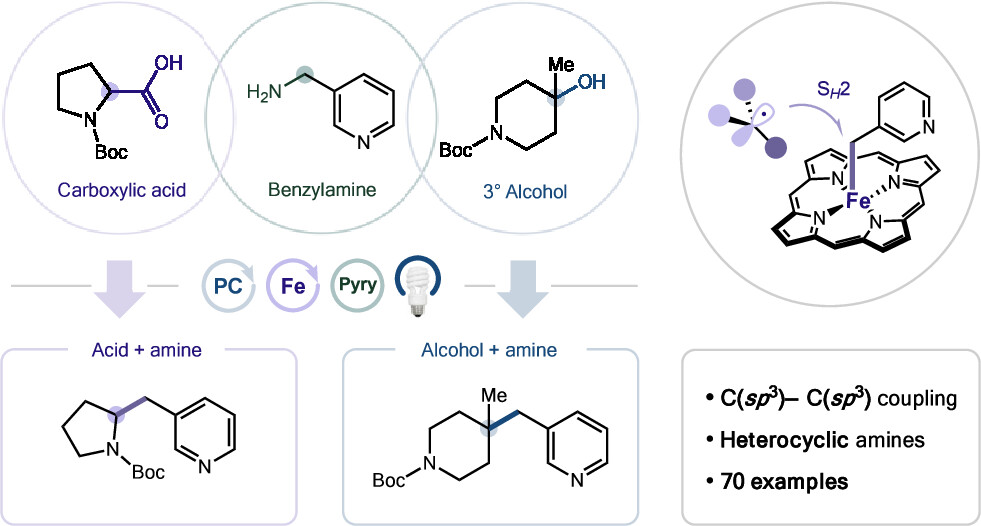
Radical Sampling Enabled Saturated N-Heterocycle Cyclization
Q. Cai,† N. B. Bissonnette,† S. Kim, T. Knauber, G. M. Chinigo, D. C. Blakemore & D. W. C. MacMillan*
J. Am. Chem. Soc. 2026, ASAP (DOI: 10.1021/jacs.6c01294) 🔓

Nitrogen-containing saturated heterocycles are key motifs in many small-molecule drugs, and methods for their rapid synthesis remain highly desirable. A streamlined approach involves aldehyde–amine condensation followed by radical cyclization; however, existing strategies require pre-functionalized amines, limiting scope and efficiency. Here, the authors expand this method to unfunctionalized amines by using native C–H bonds as radical precursors. Selective C–H activation is achieved through radical sampling, in which reversible hydrogen atom transfer and donation steps allow radicals to equilibrate, and kinetic differences in cyclization rates favor formation of the six-membered heterocycle. This approach provides a general, modular strategy for constructing saturated heterocycles directly from aldehydes and amines.
Catalytic C-Demethylation of Phenols and Anilines Enabled by a Removable Mono-Directing Group
Y. Xue,† L. Kan,† L. Yiu, C. Yu* & G. Dong*
J. Am. Chem. Soc. 2026, ASAP (DOI: 10.1021/jacs.6c01406)
Previously: ChemRxiv (DOI: 10.26434/chemrxiv-2025-r0r7n) 🔓

The authors report a Ru-catalyzed activation of C(aryl)–C(methyl) bonds enabled by a removable mono-directing group (DG). By employing a bulky phosphinite or aminophosphine-DG that can be easily installed and removed later, a methyl group at the ortho-position of phenols and anilines can be smoothly deleted under the hydrogenolysis conditions. This reaction exhibits a broad substrate scope and excellent functional group tolerance. Mechanistic studies suggest that a Ru–hydride species generated in situ is responsible for the C(aryl)–C(methyl) bond activation, with methane identified as a by-product.
Pyridine to Pyridazine Skeletal Editing via CN-to-NN Atom-Pair Swap
M. Haring,† D. Bhattacharya,† P. Xu, D. B. Fadhla, C. G. Daniliuc & A. Studer*
J. Am. Chem. Soc. 2026, ASAP (DOI: 10.1021/jacs.6c03122)

The authors report a CN-to-NN atom-pair swap that converts pyridopyrimidones—dienamines generated by temporary dearomatization of pyridines—into pyridazines via a one- or two-pot dearomatization–[4+2]–retro-[4+2]–aromatization sequence with PTAD as the N=N dienophile. The synthetic utility of this method is demonstrated through late-stage skeletal editing of complex molecules and a 7 mmol scale reaction.
Chemoselective Reduction of Nitroarenes to Anilines Using a Nickel Foam
P. Kumar, M.-J. Tom, R. L. Grange, D. Esau, P. Miller, G. Jerkiewicz* & P. A. Evans*
J. Am. Chem. Soc. 2026, ASAP (DOI: 10.1021/jacs.5c19584)

The reduction of nitroarenes to anilines is achieved using nickel foam in acidic ethanol at room temperature. A defining feature of this protocol is its exceptional chemoselectivity, enabling selective reduction of nitro groups in the presence of aryl halides, nitriles, alkenes, alkynes, and carbonyl derivatives. This selectivity makes the method particularly powerful for preparing highly functionalized aromatic amines, key motifs in the construction of complex targets, as exemplified by the synthesis of several important drugs and natural products.
👉 C&EN write-up, here.
Confined Acids Catalyze a Broadly Applicable β-Selective O-Glycosylation
J. Lu,† T. Zheng,† S. Matsutani, N. Tsuji, C. Zhu, M. Leutzsch & B. List*
J. Am. Chem. Soc. 2026, ASAP (DOI: 10.1021/jacs.6c01167)

The development of methods for precise, stereocontrolled glycosidic bond formation has been a long-standing objective in chemistry. General catalytic β-selective glycosylations to date are largely limited to SN2-like reactions, with competing SN1-like pathways via oxocarbenium ions eroding selectivity. Here, the authors report an alternative strategy: confined acids catalyze broadly applicable β-selective O-glycosylations—including 2-deoxyglucosylation, glucosylation, and mannosylation—which mechanistic and theoretical studies indicate proceed via an SN1-like pathway.

Triazenyl Furans as Diels–Alder Dienes
J. E. Budwitz,† S. A. Hollers,† A. R. Wester & C. G. Newton*
ChemRxiv 2026 (DOI: 10.26434/chemrxiv.15001641/v1) 🔓

2-Triazenyl furans are introduced as bench-stable, electron-rich Diels–Alder dienes, enabling access to highly substituted arenes under redox-neutral conditions. The triazene substituent activates the furan toward cycloaddition, promotes in situ aromatization, and serves as a versatile handle for downstream functionalization. Diels–Alder reactions are demonstrated with a broad range of dienophiles in both inter- and intramolecular settings. Chemoselective derivatizations highlight the synthetic versatility of this platform, culminating in concise annulative syntheses of pomalidomide, (S)-apremilast, and related analogues.
Addressing Steric and Electronic Limitations in Nickel Photoredox C–N Cross-Coupling Reactions
I. Ghosh,*† J. Düker,† S. Pillitteri, A. Dellisanti, P. Feuerer, M. Philipp, T. Wild, M. J. Johansson, R. M. Gschwind, M. S. Sigman* & Burkhard König*
ChemRxiv 2026 (DOI: 10.26434/chemrxiv.15001838/v1) 🔓

Nickel–photoredox catalysis is a powerful platform for C–N cross-coupling, yet conditions that accommodate both electron-rich and sterically hindered aryl halides remain limited due to slow oxidative addition and catalyst deactivation. Here, the authors report a general system that overcomes these challenges through the synergistic combination of a bipyridine ligand and alkylamine additives. This strategy enables high-yielding C–N bond formation across a broad range of electronically and sterically demanding substrates. Mechanistic studies indicate that enhanced performance arises from dynamic ligand interactions that stabilize and reactivate off-cycle nickel species. Multivariate linear regression analysis reveals that electronic effects contribute approximately twice as much as steric effects to oxidative addition kinetics.

Radical Deoxygenation of Alcohols via Visible Light Irradiation of a Titanium Porphyrin Catalyst
B. M. Gross, S. Razavi, A. J. de Almenara & B. M. Stoltz*
Org. Lett. 2026, ASAP (DOI: 10.1021/acs.orglett.6c00955) 🔓

Alcohols are ubiquitous motifs in organic molecules that can be employed as radical precursors, usually activated through a two-step process. Here, the authors present the unique ability of a titanium porphyrin complex to directly deoxygenate alcohols without pre-activation. The system generates the carbon-centered radical under mild reaction conditions and turns over the catalyst via photoexcitation with purple LEDs. To demonstrate its value, the radical intermediate can be utilized for Giese addition and nickel-mediated cross-coupling.

Transition Metal-Catalyzed Direct C(sp3)–H Functionalization Reactions of Aliphatic Carboxylic Acids
S. Mal & M. van Gemmeren*
Chem. Rev. 2026, ASAP (DOI: 10.1021/acs.chemrev.5c01044) 🔓

This review provides a comprehensive overview of transition metal-catalyzed C(sp3)–H functionalization reactions enabled by carboxylic acids, spanning contributions from 1991 to 2025. It examines the distinct mechanistic manifolds underlying these transformations, systematically comparing their reactivity, limitations, and opportunities. The discussion encompasses reaction design, substrate scope, and key mechanistic features, alongside a critical assessment of current methodologies, offering insights to guide future developments in the field.

Earthrise (‘68) vs. Earthset (‘26)

Earthrise (1968): Earth emerging above the Moon’s horizon, captured by Apollo 8.

Earthset (2026): Earth slipping behind the Moon’s horizon, captured by Artemis II.
🌕 / 🌑 Earthrise (‘68) vs. Earthset (‘26). Nearly six decades after humanity’s first journey around the Moon, last week marked our most recent return to lunar orbit. During its fly-by on 6th April, the Artemis II crew travelled nearly half a million kilometres from Earth (the farthest humans have ever ventured) capturing incredible images and offering a rarely seen perspective of deep space. Among the standout images is a striking “Earthset”: a crescent Earth slipping behind the Moon’s horizon. The image parallels the iconic “Earthrise” photograph captured by Apollo 8 astronauts, but in reverse, showing Earth disappearing rather than emerging.
As the spacecraft swept ~6,500 km above the surface, the crew also observed: a nearly hour-long solar eclipse, subtle colour variations including browns and hints of green across the 930 km-wide Orientale basin, and at least five meteoroid impact flashes—brief bursts of light caused by micrometeoroids striking the lunar surface.
While the mission gathered data on deep space radiation exposure, the crew also proposed names for two previously unnamed lunar craters: “Integrity”, after their spacecraft, and “Carroll”, in tribute to Commander Reid Wiseman’s late wife. “It’s a bright spot on the Moon, and we would like to name it Carroll”, said mission specialist Jeremy Hansen. In a link to the 1968 fly-by, the astronauts also received a pre-recorded message from Apollo 8 pilot Jim Lovell, who passed away last year. Excerpt below:
“Welcome to my old neighbourhood! […] Don’t forget to enjoy the view”.
That’s all for this issue! Have a great week and we’ll see you next Monday.