Monday 30th March – Sunday 5th April 2026
Volume 3, Issue 13


SuzukiMe: A Tactically Mild Methylation Reagent
R. C. Harbit,† L. P. M. de Oliveira Leão,† S. V. Korobko, S. O. Sotnik, D. M. Volochnyuk, S. V. Ryabukhin & Joel M. Smith*
ChemRxiv 2026 (DOI: 10.26434/chemrxiv.15001572/v1) 🔓

The authors report a mild and practical method for methylating aryl bromides and triflates using activated boronate nucleophiles. A pre-activated lithium dialkyl boronate salt enables highly reactive and selective B-alkyl Suzuki coupling without exogenous base, improving tolerance to sensitive substrates. The method shows broad scope, including late-stage methylation of complex molecules, and is applicable to isotopic labeling.

Divergent and Programmable Skeletal Remodeling of Complex Macrocycles with a Small Method Set
A. Nikbakht,† X. Li,† J. Wan, C. Qin & A. H. Hoveyda*
Science 2026, First Release (DOI: 10.1126/science.aee3540)

The authors report a programmable strategy for skeletal remodeling of macrocycles, enabling systematic modification of ring size and structure from a common diene or macrocyclic olefin “hub”. A small set of catalytic transformations allows iterative ring expansion, contraction, and functionalisation under mild conditions. The approach provides streamlined access to diverse macrocyclic analogues, exemplified by the synthesis of 14 epothilone C derivatives in an average of three steps per analogue. This platform offers a practical alternative to de novo synthesis for exploring macrocycle structure–activity relationships.

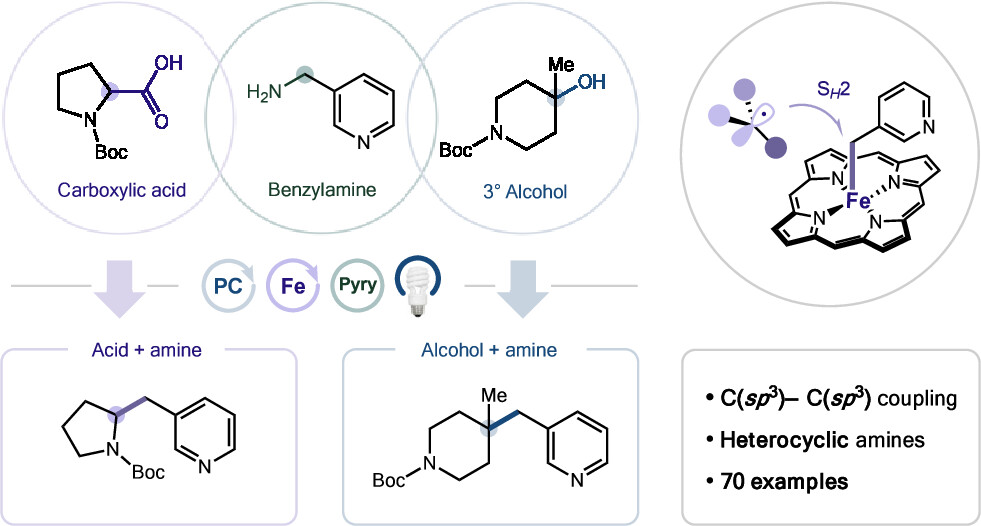
The Synthesis of Tertiary Alkylamines from Alkyl-Substituted Alkenes Using Amine Umpolung Strategy
S. Nakatsuka,† T. Kinoshita,† Y. Sakakibara* & K. Murakami*
J. Am. Chem. Soc. 2026, ASAP (DOI: 10.1021/jacs.6c02320) 🔓

The authors report a radical aminomethylation of unactivated alkenes enabled by α-ammoniomethyl radicals as electrophilic umpolung equivalents of α-aminomethyl radicals. Under visible-light photoredox conditions, this strategy achieves regioselective difunctionalisation of a broad range of aliphatic alkenes, overcoming longstanding polarity constraints. A one-pot protocol furnishes highly substituted tertiary alkylamines with broad functional-group tolerance, and extends to allylamine synthesis and diverse SOMOphile coupling.
Excited-State SNAr Reactions of Nitroarenes
Z. Lyu,† T. Liang,† G.-J. Cheng* & F. Ye*
J. Am. Chem. Soc. 2026, ASAP (DOI: 10.1021/jacs.5c21841)

The authors report an excited-state SNAr reaction of nitroarenes under visible light irradiation, where triplet-state electronic reorganisation enables CAr–NO2 substitution via pathways inaccessible in the ground state. Guided by computational analysis, a practical protocol for denitrative substitution across diverse polycyclic arenes is developed.
Copper-Catalyzed Enantioconvergent Nucleophilic Fluorination of Alkyl Electrophiles to Generate α-Fluoroamides
Z.-Y. Wang & G. C. Fu*
J. Am. Chem. Soc. 2026, ASAP (DOI: 10.1021/jacs.6c03178) 🔓

The authors report the catalytic asymmetric synthesis of alkyl fluorides through copper-catalyzed enantioconvergent substitutions of racemic alkyl halides, specifically, fluorinations of α-haloamides by CsF, enabling carbon–fluorine bond formation with good enantioselectivity and yield under mild conditions. This new process was applied to a streamlined, catalytic asymmetric synthesis of a bioactive α-fluoroamide and to the synthesis of other classes of enantioenriched fluorinated compounds.
Photocatalytic, Regioselective Enone Deconjugation: An Enabling Platform for Terpene Modification
L. Blank, C. Mück-Lichtenfeld, C. G. Daniliuc & R. Gilmour*
J. Am. Chem. Soc. 2026, ASAP (DOI: 10.1021/jacs.6c02442)

The authors report a photocatalytic strategy for alkene relocation via regioselective deconjugation of cyclohexenones, inspired by steroid Δ5-isomerase. A traceless C(sp3)–Br handle enables controlled generation of a key photodienol intermediate, favouring endo-selective isomerisation pathways inaccessible under ground-state conditions. The method operates under mild visible-light conditions and enables access to β-isophorone derivatives and 3-oxo-Δ5-steroids, valuable motifs in industrial and pharmaceutical settings.
Diastereoselective Synthesis of Housanes via the Carbocupration of Cyclopropenes
C. Tanguy, V. Smyrnov, M. D. Wodrich & J. Waser*
J. Am. Chem. Soc. 2026, ASAP (DOI: 10.1021/jacs.6c03013)
Previously: ChemRxiv (DOI: 10.26434/chemrxiv.10001480/v1) 🔓

Strained bicyclic frameworks are valuable isosteres, but the bicyclo[2.1.0]pentane (“housane”) scaffold remains synthetically challenging with limited substitution patterns. Here, the authors report a diastereoselective carbocupration–cyclization cascade of homocarbonyl cyclopropenes that provides efficient access to diverse housanes. By exploiting carbonyl groups as dual-purpose directing groups and electrophiles, the method achieves high atom and step economy. The resulting scaffold mimics 1,3-cyclopentanes and undergoes controlled strain-driven ring-opening, supporting its use as an isostere or covalent binder. The cascade also generates an alcohol handle for diversification, enabling the first high-yielding synthesis and isolation of bicyclo[2.1.0]pentanone (“housone”).
Total Synthesis of Ussuriedine via a Late-Stage Stevens Rearrangement: Implications for Biosynthesis
D. Baidilov,† K. J. Cassaidy,† Y.-J. Shin & V. H. Rawal*
J. Am. Chem. Soc. 2026, ASAP (DOI: 10.1021/jacs.6c00926) 🔓

The authors report the first total synthesis of ussuriedine, a heptacyclic steroidal alkaloid featuring an unprecedented octahydro-3,6-methanoquinolizine framework. A convergent strategy couples decalin and piperidine fragments, enabling an intramolecular [2+2+2] cyclotrimerisation to construct the core scaffold. In the endgame, nitrogen quaternisation and a [1,2]-Stevens rearrangement forge the final ring, completing the natural product in 23 steps. The synthetic design provides insight into the potential biosynthetic origin of this complex architecture.
Asymmetric Total Synthesis and Structure Revision of (+)-Mangicol D
Y. Xie, Y. Wang, Z. Zhu, S. Ning & T. J. Maimone*
J. Am. Chem. Soc. 2026, ASAP (DOI: 10.1021/jacs.6c01185)

Isolated from the marine fungus Fusarium heterosporum, mangicols are a class of sesterterpenoids featuring unprecedented spirotetracyclic cores. Here, the authors report the first asymmetric synthesis of a mangicol sesterterpene using a domino carbopalladation–carbonylation, Conia–ene cyclization, and SmI2-mediated reductive cyclization to efficiently construct the spiro-fused tetracyclic framework. These results support a stereochemical revision of the polyol side chain in the originally assigned mangicols.

Propellane Alkaloid Biosynthesis and Total Synthesis via Interrupted Reaction Pathways
J. M. Billingsley,† J. Ding,† A. T. Hands, K. Niwa, N. J. Adamson, L. A. Wein, B. Perlatti, N. K. Garg* & Y. Tang*
ACS Cent. Sci. 2026, ASAP (DOI: 10.1021/acscentsci.6c00057) 🔓

This study reports the biosynthesis of subrubine alkaloids, a novel family of fungal pyrrolidinoindolines with a congested diaza[3.3.3]propellane core, alongside the total synthesis of pensubrubine. Genome mining identified the noncanonical ene-reductase SubF as a propellane synthase, rerouting an enolate intermediate toward an intramolecular Mannich cyclization. Complementing the biosynthetic work, a concise 7-step total synthesis of pensubrubine was developed using a diastereoselective interrupted Fischer indolization to construct the azapropellane framework and establish its absolute configuration.

Late-Stage Oxygenation in Organic Synthesis: From Innate Reactivity to Directed Selectivity in Complex Settings
K. Gennaiou, M. Kourgiantaki, K. Mazaraki, F. Richard, N. Fincias, S. Arseniyadis* & A. L. Zografos*
Angew. Chem. Int. Ed. 2026, Early View (DOI: 10.1002/anie.202521362)

Late-stage oxygenation (LSO) enables the selective installation of oxygen functionality in complex molecules at advanced synthetic stages, offering powerful opportunities for molecular diversification. Recent advances in selective oxidants, biocatalysis and photoredox methods have significantly expanded the scope and precision of LSO, with growing impact in pharmaceutical, agrochemical and materials science. This Review outlines the key conceptual and mechanistic foundations of LSO, alongside emerging chemical and enzymatic strategies, providing a practical guide to predictable oxygenation in complex settings.

O-Vinylhydroxylamines for de novo Pyrrole Synthesis
C. Randolph, O. Buravov, Z. Grimm, P. K. Mykhailiuk* & L. Kürti*
ChemRxiv 2026 (DOI: 10.26434/chemrxiv.15001468/v1) 🔓

The authors report that O-vinylhydroxylamines enable the de novo construction of pyrroles via an aza-Michael/oxaza-Cope [3,3]-sigmatropic rearrangement/aromatization cascade with activated alkynes. This one-pot transformation proceeds rapidly at room temperature under mild base catalysis, furnishing dihydropyrrole hemiaminal intermediates that can be directly converted to pyrroles or selectively functionalised. The method exhibits broad scope and provides access to substitution patterns that previously required harsh conditions or hazardous acetylene-based methods.
Carbonylative Aminative Suzuki–Miyaura Coupling: Pd-Catalyzed Synthesis of Amides from Vinyl/Aryl Halides and Boronic Acids
J. Zhou,† D. Raykowski,† Z. Kowalski, S. Li, Y. Ping, Y. Luo, E. E. Kwan & R. Y. Liu*
ChemRxiv 2026 (DOI: 10.26434/chemrxiv.15001482/v2) 🔓

The authors report a four-component variant of the Pd-catalyzed Suzuki–Miyaura coupling that repurposes partners conventionally used for C–C bond formation to instead yield amides. The sequential insertion of CO, from Mo(CO)6 as a convenient surrogate, and NH, from O-(diphenylphosphinyl)hydroxylamine (DPPH), allows access to a broad scope of amides, enamides, α,β-unsaturated amides, and α,β-unsaturated enamides. Drug-like analogs can be rapidly assembled and “reverse amide” counterparts can be generated from the same starting materials.
Unique Fluoroalkyl Substituents: The Case of Fluoroisopropyl (–CFMe2) Group
V. Sham, S. Fok, A. Rushchak, M. Komashnia, D. Lutfullin, D. Kyslyi, Y. Makovetska, M. Mitiuk, A. Dudnyk, A. Pochepko, Y. Galushchak, V. Konashuk, O. Stanko, A. Rudnichenko, D. Skrypnik, A. Tiasko, O. Osypenko, O. Yakymchuk, J. Adamczyk, P. Borysko, V. Kubyshkin, I. Sadkova & P. K. Mykhailiuk*
ChemRxiv 2026 (DOI: 10.26434/chemrxiv.15001396/v1) 🔓

The CFMe2-substituent is a close analogue of the CF3-group. The CF3 for CFMe2 replacement in model compounds mostly does not affect key physicochemical properties: water solubility, lipophilicity and metabolic stability. Analogous replacement in the anticancer drug Enasidenib, however, provides a biologically active analogue with improved solubility and reduced lipophilicity.

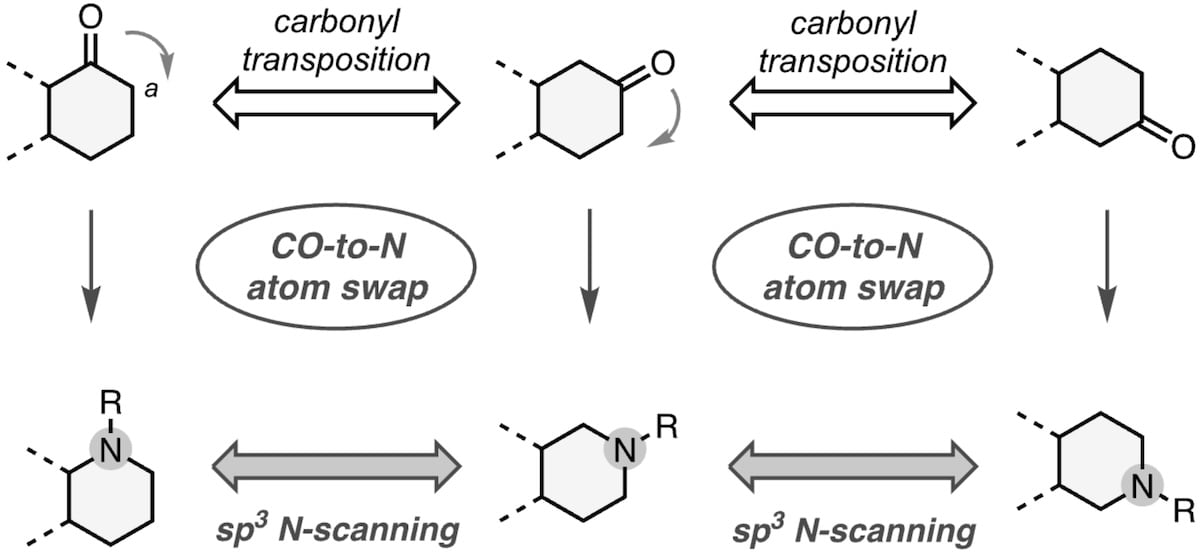
Oxygen-Atom Replacement in Non-Strained Cyclic Ethers
Y.-Q. Zhang, K. Z. Yap, S.-H. Li, F. W. X. Lim & M. J. Koh*
Chem 2026, Online Now (DOI: 10.1016/j.chempr.2026.103000)

The authors report a phosphine-mediated strategy for chemoselective oxygen-to-X (X = nitrogen, sulfur, selenium or carbon) transmutation, converting non-aromatic furans and pyrans into diverse heterocyclic and carbocyclic analogues under mild conditions. The method enables late-stage editing of O-heterocyclic pharmaceutical building blocks, providing streamlined access to saturated scaffolds without the need for de novo synthesis.

One Plant, Five Psychedelics
🍃 One Plant, Five Psychedelics. In the most exciting news for the tobacco plant since tomacco, researchers have successfully engineered it into a biosynthetic powerhouse capable of producing multiple psychedelic molecules typically found across plants, fungi and animals.
In Science Advances, the authors report the first complete elucidation of the DMT biosynthetic pathway in hallucinogenic plant species, revealing a simple two-enzyme sequence: a tryptophan decarboxylase and a highly promiscuous N-methyltransferase. By expressing these enzymes in tobacco, which has a naturally abundant tryptophan pool, the team achieved de novo DMT production.
Building on this, the researchers introduced additional biosynthetic enzymes to reconstruct pathways to DMT (plants), psilocybin/psilocin (fungi), and bufotenin and 5-MeO-DMT (toads) within the same tobacco plant. The system can also generate new-to-nature compounds: the addition of bacterial halogenases enables the production of halogenated tryptamines, while AlphaFold-guided enzyme engineering delivered a ~40-fold increase in 5-MeO-DMT biosynthesis.
The work could help enable new routes for the development and manufacture of psychedelic-based therapies for depression, anxiety and PTSD, as well as more sustainable alternatives to extraction from natural sources.
That’s all for this issue! Have a great week and we’ll see you next Monday.