- Synthesis Spotlight
- Posts
- Branching Out
Branching Out
💡 The Chemical Industry & Geopolitics

Monday 16th March – Sunday 22nd March 2026 | Volume 3, Issue 11 |


Cross- and Branched-Selective Hydroalkenylation by Metal Hydride Selection
C. Li,† X.-c. Gan,† Y. Irie, M. A. Smith & R. A. Shenvi*
Science 2026, 391, 1262–1268 (DOI: 10.1126/science.aeb2389)
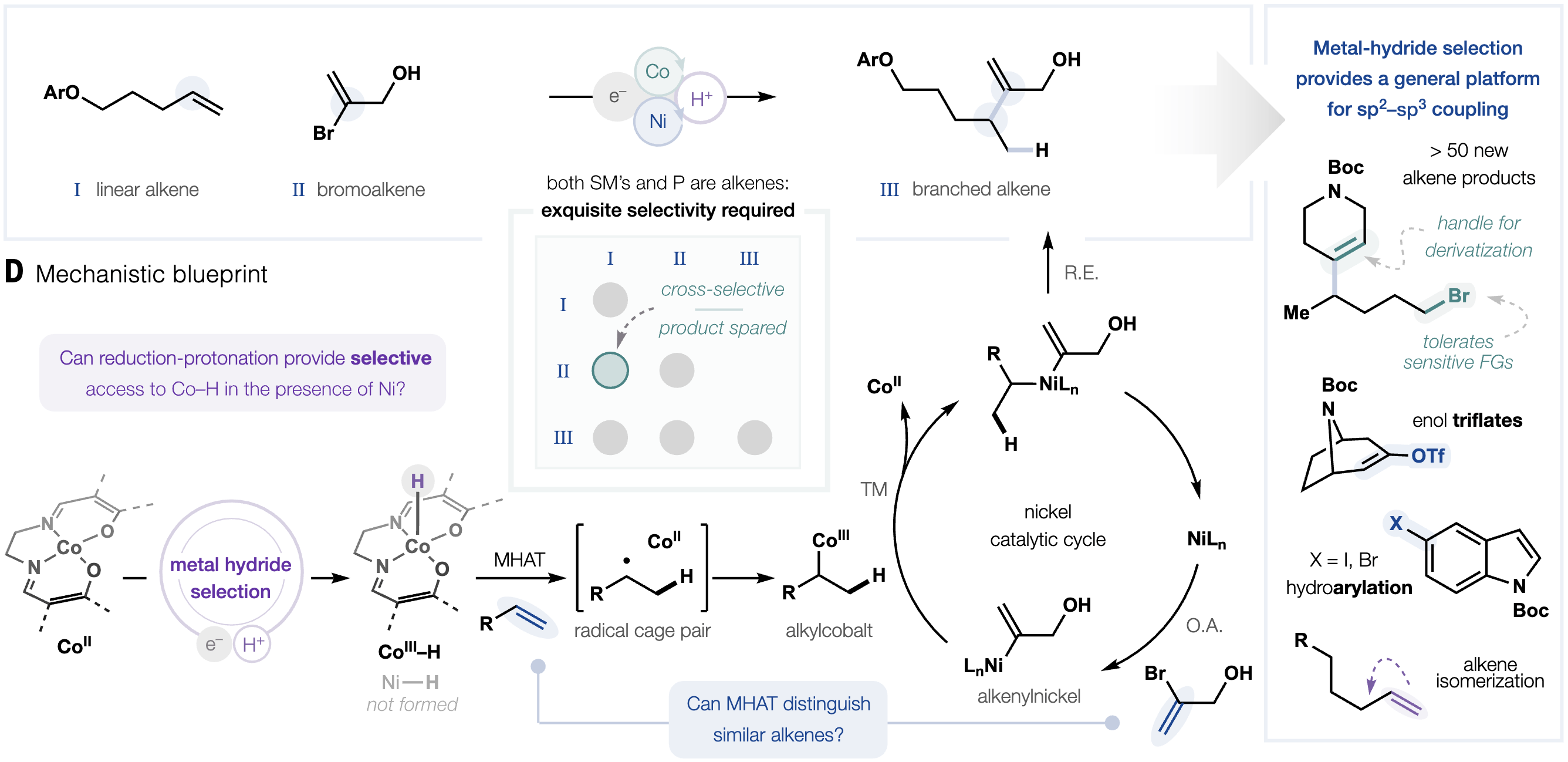
Controlled placement of branch points along carbon chains is essential in the synthesis of materials, agrochemicals and pharmaceuticals. Metal hydride hydrogen atom transfer (MHAT) to alkenes enables direct access to branched products from abundant α-olefins and can be merged with transition-metal catalysis to override inherent linear selectivity. However, poor discrimination of hydridic reagents between catalysts often generates multiple metal hydrides, leading to diminished selectivity. Here, the authors introduce a “metal hydride selection” strategy in which a lutidinium acid/manganese system selectively forms cobalt hydrides in the presence of nickel. This enables a cross- and branched-selective hydroalkenylation of alkenes, delivering valuable branched products with high selectivity.

Direct Conversion from Alkenes to Alkynes
J. Meng, Y. Liang, R. Xu, Z. Cheng, Y. Huang, H. Shi, Y. Chen, X. Wang, J. Wei, T. Wang, B. Zhao & N. Jiao*
Nature 2026 (DOI: 10.1038/s41586-026-10372-3)
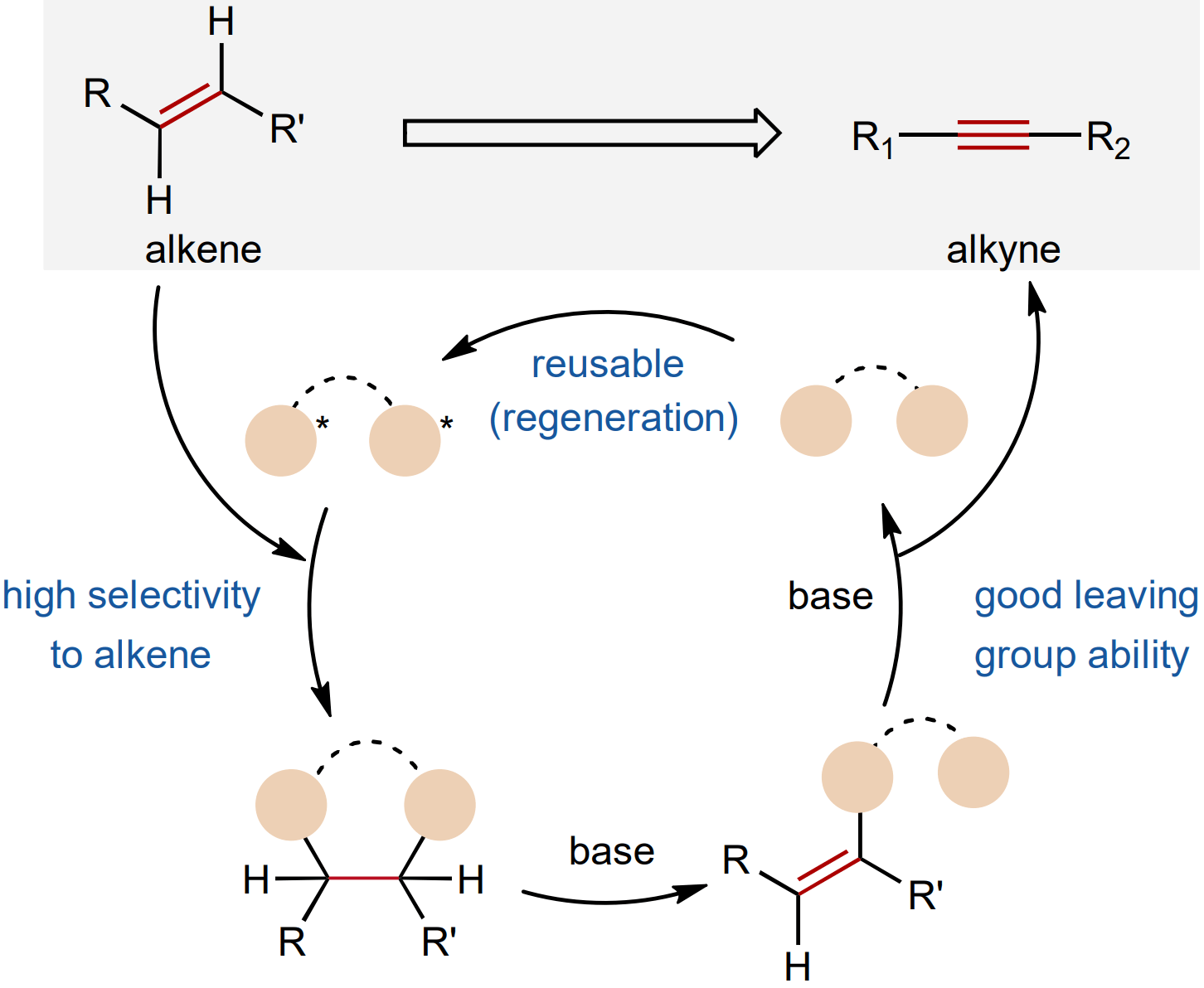
The authors report a recyclable selenanthrene reagent that enables the mild desaturation of alkenes to alkynes. The method exhibits broad compatibility with both classical leaving groups and sensitive functional groups, facilitating late-stage application in the synthesis of complex alkynes. Additionally, the platform allows Z/E alkene configuration inversion or sorting, which is not accessible using existing methods.

Electroreductive Cleavage of C(sp3 )–N Bonds in Saturated N-Carbonyl Heterocycles
R. Buscemi,† P. Martínez-Balart,† D. Bura,† M. Díaz-Ruiz,† J. Moreno-González, E. M. C. Pinheiro, J. J. Douglas, C. Trujillo* & G. E. M. Crisenza*
J. Am. Chem. Soc. 2026, ASAP (DOI: 10.1021/jacs.5c21454)
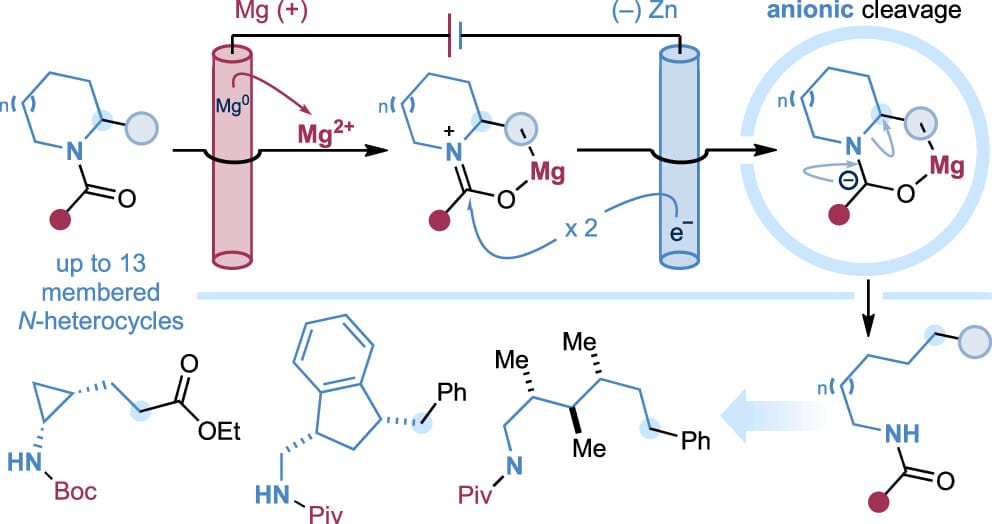
The authors report an electroreductive strategy for ring-opening C–N bond cleavage of saturated N-carbonyl heterocycles to access linear amides. This method enables heterolytic ring opening across a broad range of unstrained cyclic amines, including pyrrolidines, piperidines, azepines, azocanes, and N-macrocycles. Broad compatibility with diverse N-carbonyl protecting groups and functional groups is demonstrated, and the procedure is readily scalable and amenable to high-throughput experimentation. The synthetic utility of the method is demonstrated through the preparation of decorated, stereodefined linear amides from complex cyclic precursors.
Evolution of a Synthetic Strategy for Complex Diterpenes from Euphorbiaceae and Thymelaeaceae
K. Yu, V. H. Vasilev, L. Spessert & T. J. Maimone*
J. Am. Chem. Soc. 2026, ASAP (DOI: 10.1021/jacs.5c22127) 🔓
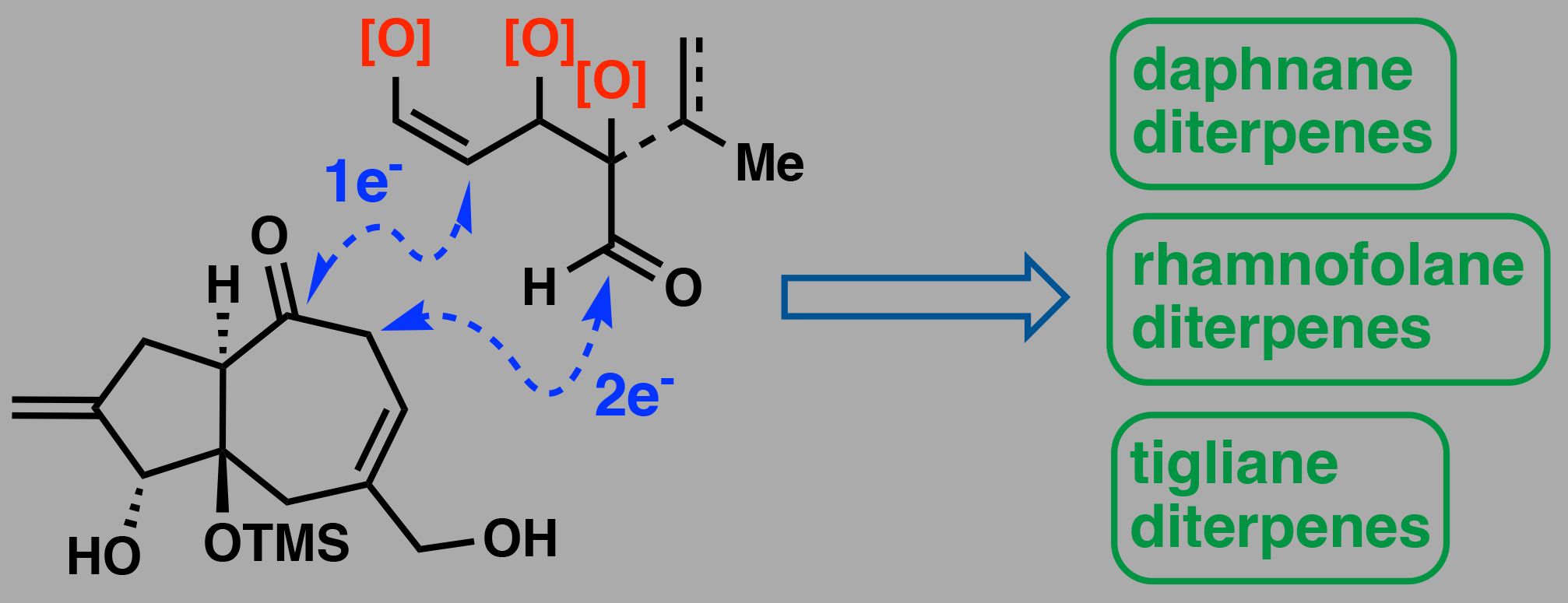
The biosynthetically related daphnane, tigliane, and rhamnofolane diterpenes comprise a structurally complex class of natural products with significant therapeutic potential. Despite decades of study, many members remain synthetically inaccessible due to their intricate 5,7,6-fused tricyclic cores and varied oxidation patterns. Here, a convergent synthetic strategy based on two modular fragments is described, the evolution of which enabled the total synthesis of the flagship daphnane diterpene orthoester resiniferatoxin. Furthermore, adaptation of this approach provided access to a common tricyclic ketone intermediate, thus achieving formal syntheses of rhamnofolane and tigliane diterpenes, including crotophorbolone, phorbol, and prostratin.
3,5-Dimethylorsellinic Acid (DMOA)-Derived Meroterpenoid Ortholactones (−)-Novofumigatonin, (−)-Asnovolin F, and (−)-Asnovolin G: Total Syntheses and Related Studies
L. J. Sprenger,† V. A. P. Ruf,† K. Volynskiy, V. M. Kandler, N. Nasiri, D. Martymianov & E. M. Carreira*
J. Am. Chem. Soc. 2026, ASAP (DOI: 10.1021/jacs.6c01821)
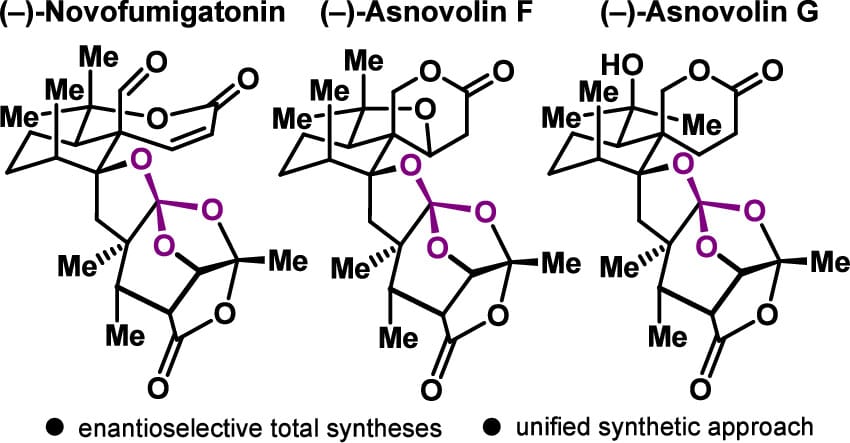
The authors report a full account of the total synthesis of (−)-novofumigatonin and the first syntheses of (−)-asnovolin F and (−)-asnovolin G. The strategy for these 3,5-dimethylorsellinic acid (DMOA)-derived spiromeroterpenoid ortholactones relies on two key elements: convergent coupling of fragments of comparable complexity and subsequent construction of the embedded ortholactone cages. An advanced intermediate containing the ortholactone–acetal core enables access to all three natural products.
Stereodivergent Construction of trans-Decalin-Based Terpenoids
S. L. Barlock, A. S. Shved, K. D. Landers, B. K. Mai, S. Fujiki, P. C. Ryffel, W. Y. Lo, A. G. Feng, N. W. Wade, L. Zhu, V. D. Nguyen, D. A. Petrone,* S. E. Denmark* & D. Sarlah*
J. Am. Chem. Soc. 2026, ASAP (DOI: 10.1021/jacs.6c01881)
Previously: ChemRxiv (DOI: 10.26434/chemrxiv-2025-8r6bx) 🔓
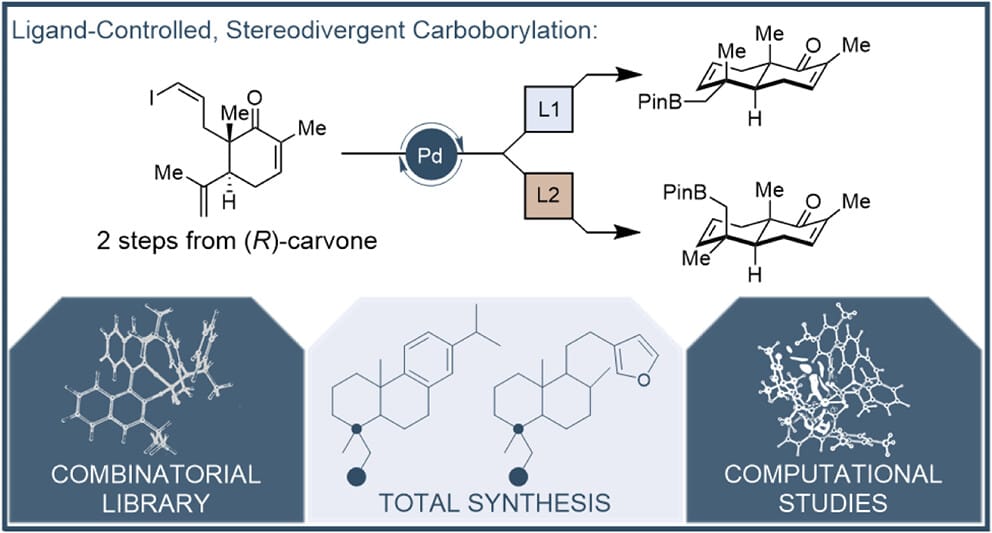
The authors report an expedient route to a versatile terpenoid building block primed for elaboration into numerous complex natural products. The intermediate is prepared from (R)-carvone, a cheap and abundant chiral pool material, in three steps via a diastereodivergent intramolecular carboborylation reaction. Notably, this method provides either equatorial or axial functionalized products from a single common precursor. Identification of optimal ligands required extensive screening, facilitated by high-throughput experimentation and an algorithmically guided in silico library, which identified the highest-performing axial-selective ligand. The synthetic utility of the method is demonstrated in the total synthesis of several diterpenoid natural products.
The RAvIN Platform: A Noncarbohydrate Platform for Oligonucleotide Synthesis
M. Nodwell,† G. Caballero-García,† J. M. Mesa,† T. Muilu, D. M. Wilson, A. Arora, A. J. Brooke, B. Singgh, M. Gill, H. M. Tran, S. M. Silverman, L.-C. Campeau, M. J. Damha & R. Britton*
J. Am. Chem. Soc. 2026, ASAP (DOI: 10.1021/jacs.5c21938)
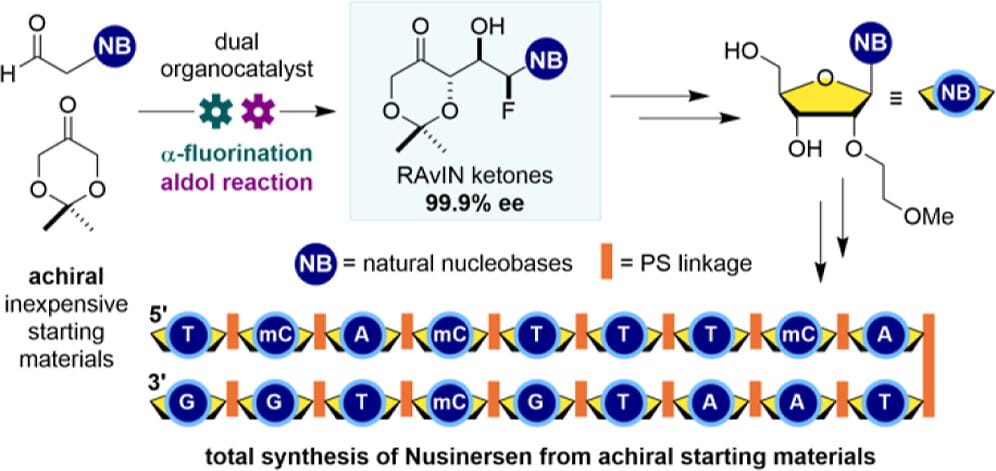
The authors report a streamlined platform for producing nucleoside analogues (NAs) to expand access to precision-edited oligonucleotide therapeutics (ONTs). The method employs achiral starting materials and a dual organocatalyst, one-pot process to generate RAvIN (rapid access to value-added innovative nucleosides) ketones (RKs), a key class of ONT building blocks. These intermediates incorporate protected natural nucleobases or analogues, can be synthesized on scale in 2–4 steps with high enantiomeric purity, and are readily diversified to access next-generation NAs with improved pharmacokinetic properties. The versatility of the approach is demonstrated through a fully noncarbohydrate synthesis of the all-MOE-modified 18mer ONT Nusinersen.

Alkoxysulfonium Salts Unlock Access to New Aryl Sulfonium Salts for Cross-Coupling
R. E. Brown, N. Kaur, A.-C. M. A. Nassoy, C. Romano & D. J. Procter*
Angew. Chem. Int. Ed. 2026, Early View (DOI: 10.1002/anie.202525298) 🔓
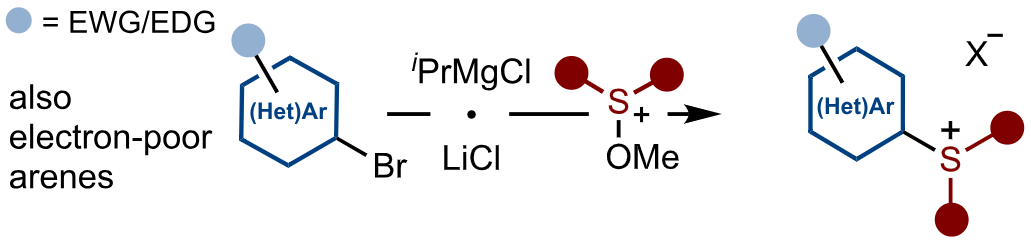
Hard-to-access (hetero)aryl sulfonium salts are prepared using stable methoxysulfonium salts in combination with the magnesiation of (hetero)aryl bromides. The approach allows access to aryl sulfonium salts bearing electron-deficient (hetero)aryl rings, or electron-rich aryl motifs in which the sulfonium motif resides at an alternative position on the ring to that delivered by electrophilic sulfenylation. The new sulfonium salts extend the scope of photocatalytic, photochemical, and ligand-coupling processes.

Innate Chiroselectivity in the Formation of Pabmaragramin, a Scalemic Natural Product
A. Gu, C.-C. Tseng, M. Pocheć, R. Szabla* & A. L. Lawrence*
ChemRxiv 2026 (DOI: 10.26434/chemrxiv.15000984/v1) 🔓
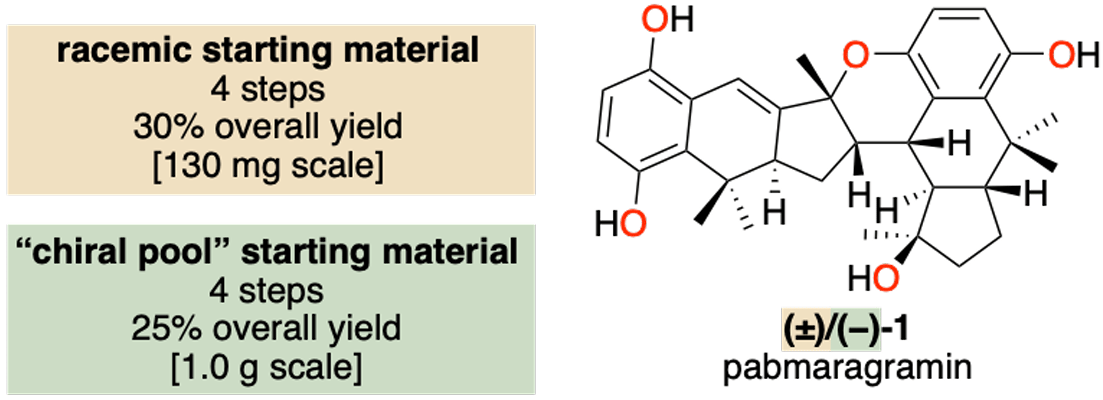
Natural products are a vital source of bioactive compounds, with activity governed by their three-dimensional structure and absolute configuration. While enzymatic biosynthesis typically affords enantiopure products and nonenzymatic pathways yield racemates, scalemic natural products challenge this dichotomy. One such example is pabmaragramin, a metabolite of Marasmius graminum isolated in a 65:35 enantiomeric ratio. Here, the authors report a four-step total synthesis of pabmaragramin, enabling access to both racemic and enantioenriched material. The route features a biomimetic cascade incorporating a Diels–Alder dimerization with inherent homochiral selectivity; computational studies provide insight into the origins of this rare selectivity.
General Access to C-Glycosides via Redox-Neutral Radical Cross-Coupling of Glycohydrazides
Y. Guo,† Y. Li,† B. Buchberger,‡ Y. Liu,‡ C. Capone, T. Adak, S. Ojha, J. L. Tyler, P. Neigenfind, M. Nassir, Y. Kawamata,* V. K. Aggarwal* & P. S. Baran*
ChemRxiv 2026 (DOI: 10.26434/chemrxiv.15001005/v1) 🔓
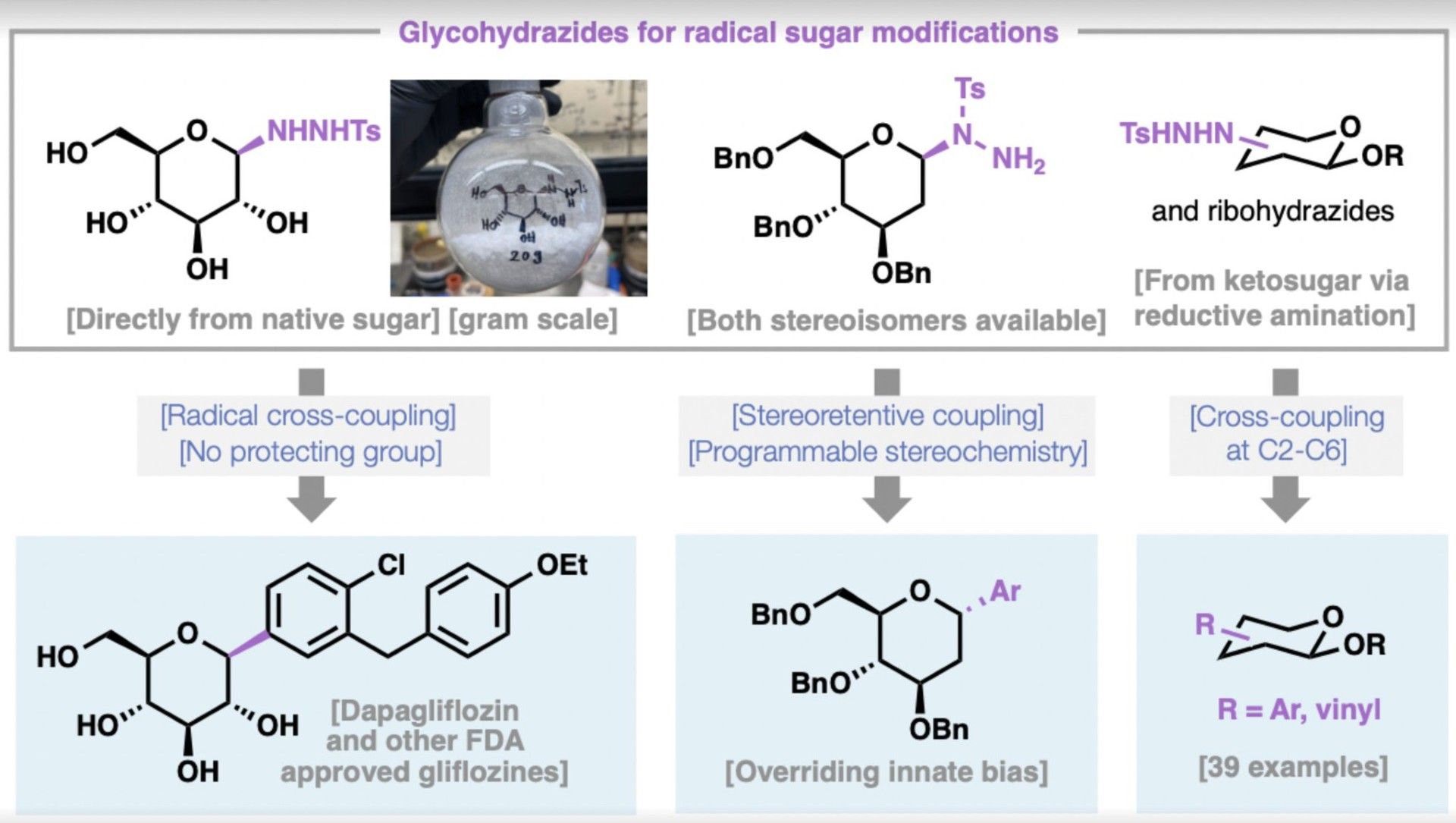
A practical approach to C-aryl glycosides using glycosyl sulfonyl hydrazides as redox-neutral radical precursors for cross-coupling is described. Prepared directly from unprotected native sugars, these reagents generate glycosyl radicals under mild conditions and enable efficient access to diverse C-aryl glycosides, including all approved SGLT2 inhibitors (e.g., dapagliflozin, canagliflozin and empagliflozin), as well as natural products including salmochelins and neopetrosins. Beyond anomeric functionalization, this platform enables C–C bond formation at multiple positions on carbohydrate scaffolds and supports stereoretentive radical coupling that can override inherent stereochemical biases.
Photo-Stitching of Nitroalkanes via Cross-Denitrative Olefination
J. K. Mitchell,† S. P. Desai,† J. M. Bergen, A. Reyes, M. S. Mclean, P. Ghosh, A. H. Bansode & M. Parasram*
ChemRxiv 2026 (DOI: 10.26434/chemrxiv.15001045/v1) 🔓
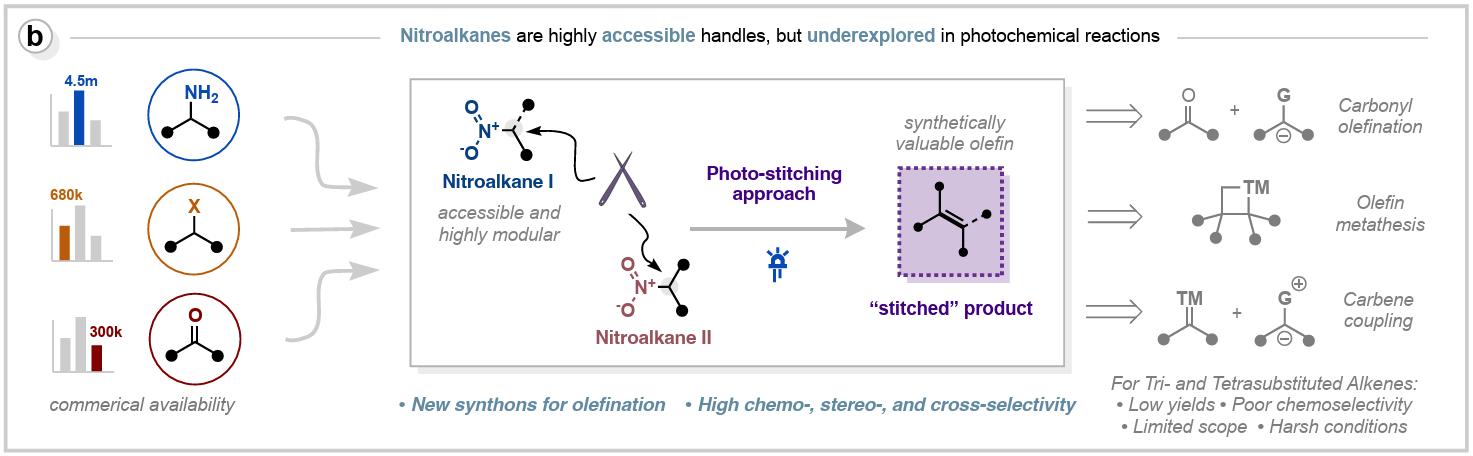
Stereodefined tri- and tetrasubstituted alkenes remain challenging to access because most olefination methods favor lower substitution and fail under sterically congested conditions. Here, the authors describe a photoinduced cross-denitrative olefination of nitroalkanes to afford tri- and tetrasubstituted alkenes. Visible-light excitation of in situ–generated nitronates promotes a photo-stitching event that couples and denitrates two nitroalkanes. The transformation exhibits broad functional-group tolerance, complements metathesis and carbonyl olefination, and scales efficiently.
👉️ For recent complementary methodology on the “photocatalytic denitrative homo- and cross-coupling of nitroalkanes towards the synthesis of alkenes” by O. Reiser and co-workers, see here.
Bicyclo[2.1.1]hexanes: Saturated Bioisosteres of meta-Benzene
A. Denisenko, Y. Makovetska, O. Stashkevych, V. Kokhalskyi, O. Shablykin, V. Levchenko, P. Garbuz, Y. Skalenko, I. Sadkova, L. Dmytrovska, P. Borysko & P. K Mykhailiuk*
ChemRxiv 2026 (DOI: 10.26434/chemrxiv.15001067/v1) 🔓
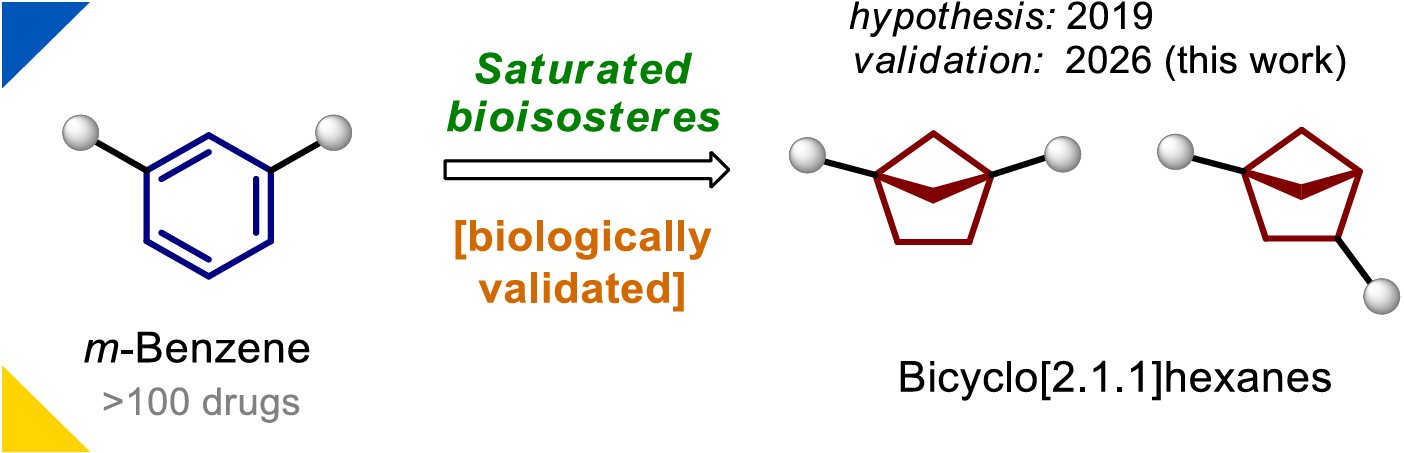
In 2019, the authors hypothesised that bicyclo[2.1.1]hexanes could serve as bioisosteres of ortho- and meta-substituted benzenes. This concept was experimentally validated for ortho-benzenes in 2020, establishing bicyclo[2.1.1]hexanes as key motifs in the emerging field of saturated benzene bioisosteres. Here, the authors provide experimental validation of the remaining hypothesis, demonstrating that bicyclo[2.1.1]hexanes can also mimic meta-benzenes.
cis-Selective Direct Halocyclopropanation of Alkenes Mediated by Nucleophilic Cobalt Photocatalysis
J. H. G. Teye-Kau, M. Pauze & S. P. Pitre*
ChemRxiv 2026 (DOI: 10.26434/chemrxiv-2025-lflm6/v2) 🔓
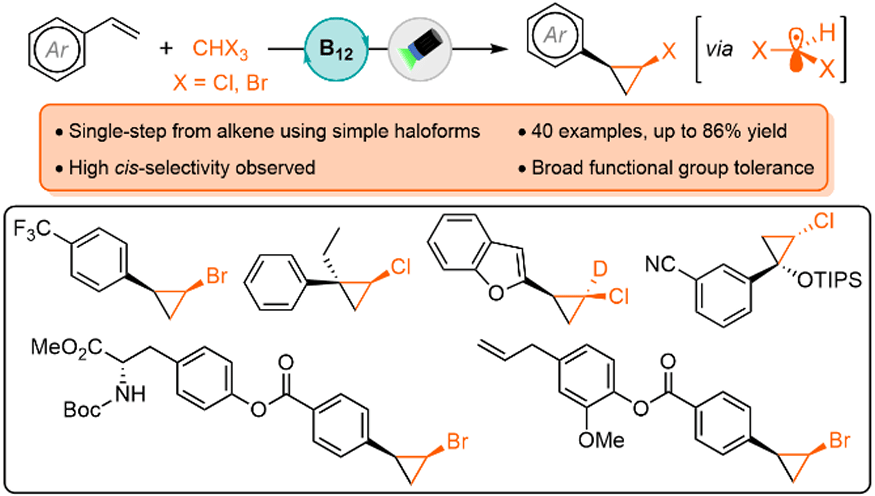
The authors report a Vitamin B12-photocatalyzed approach for the direct synthesis of 1,2-disubstituted halocyclopropanes starting from alkenes and simple haloforms under visible-light irradiation. This method yields a diverse range of 1,2-disubstituted halocyclopropanes with high cis-selectivity, providing efficient access to these traditionally difficult to prepare stereoisomers.
Collective Synthesis of Diverse Onoceroids via Chelation-Assisted Radical Cross-Coupling
H. Xu,† J. Yang,† G. Scofield, Y. Sun, A. Li,* P. Liu* & H. Renata*
ChemRxiv 2026 (DOI: 10.26434/chemrxiv.15001089/v1) 🔓
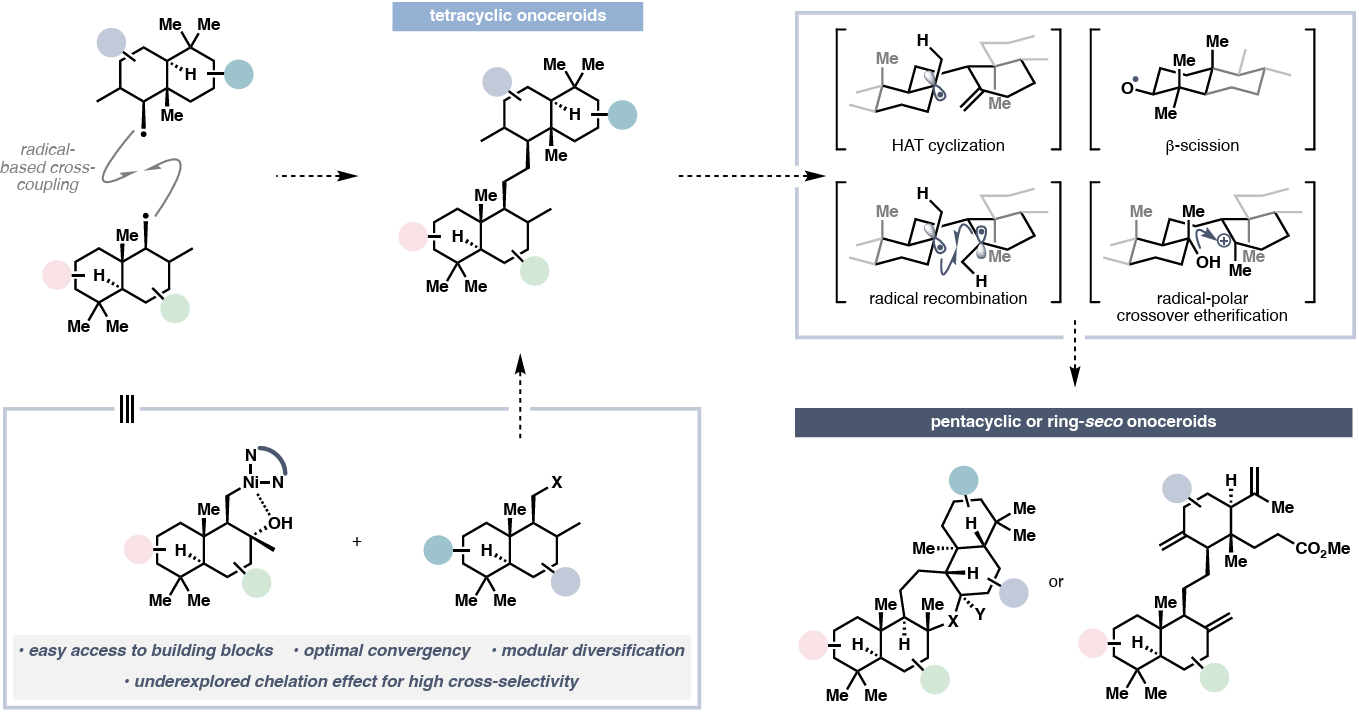
Onoceroids are complex triterpenoids that often possess unsymmetrical pseudodimeric structures, presenting a significant synthetic challenge. Here, a family-level solution to the synthesis of all known onoceroid skeleton subtypes is described. A key feature is the discovery of a chelation effect that enables high cross-selectivity in radical C(sp3 )–C(sp3 ) coupling between two closely related fragments. Combined with other modern radical reactions, this strategy enables rapid access to more than 20 naturally occurring onoceroids.

1,2-Boryl/Hydrogen Transposition via Reversible C–H Sampling
L. Xie,† P. Luo,† T. Lu,† W. Zheng,† Y. Su, J. Wang, X. Qi* & Y. Xu*
Chem 2026, Online Now (DOI: 10.1016/j.chempr.2026.102961)
Previously: ChemRxiv (DOI: 10.26434/chemrxiv-2025-lx3t5) 🔓
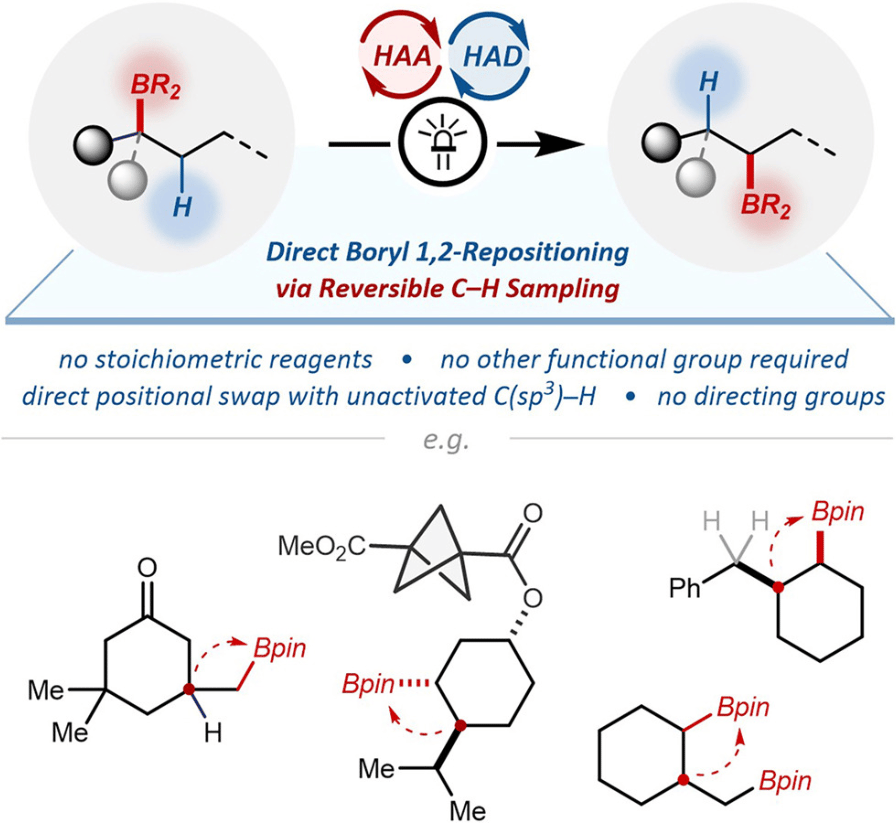
The authors report a direct 1,2-boryl translocation with common C(sp3 )–H bonds in alkyl boronic esters. Driven by the synergy between hydrogen atom abstraction and donation catalysis, this reaction features reversible radical generation at multiple C–H sites yet ensures selective transformation via the kinetically favored 1,2-radical boryl migration. No directing groups or stoichiometric reagents are needed, and the mechanism is supported by both experimental and computational evidence.

A Brønsted Acid-Base Approach for the Net Monoselective C–F Substitution of (Trifluoromethyl)alkanes
N. J. Coradi & J. S. Bandar*
Chem. Sci. 2026, Accepted (DOI: 10.1039/D6SC01042C) 🔓
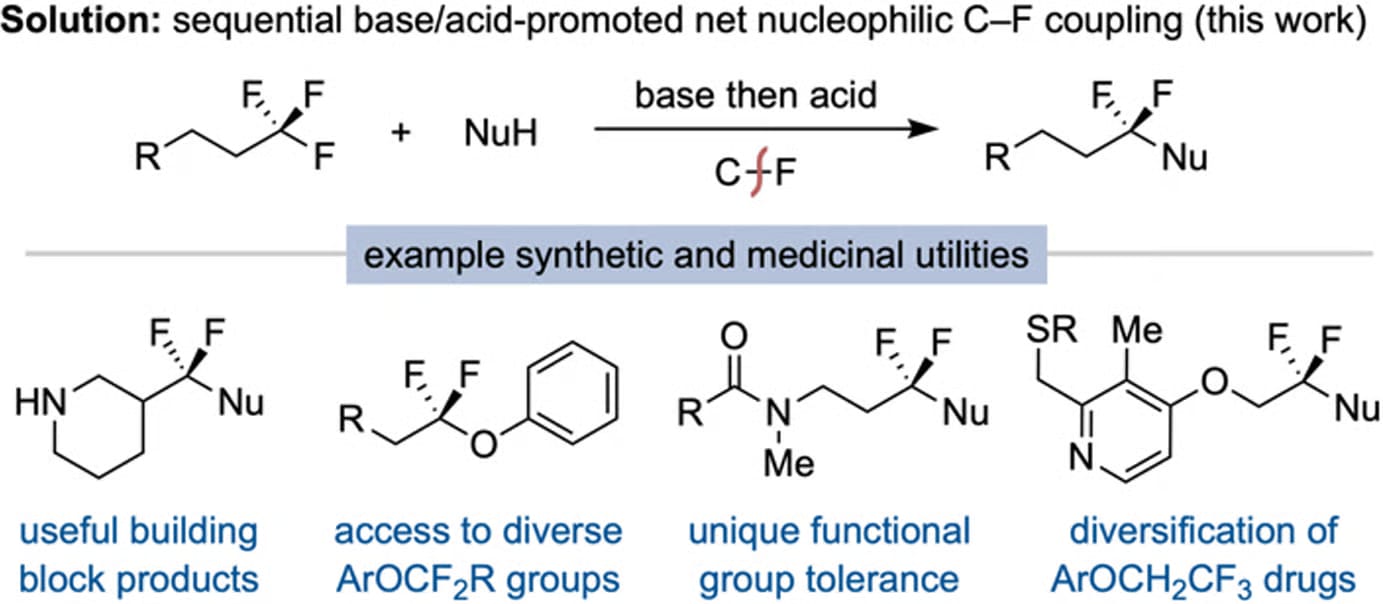
The authors disclose a method for the net coupling of nucleophiles with a single C–F bond of unactivated (trifluoromethyl)alkanes. The process proceeds through a base-promoted dehydrofluorination/defluorinative nucleophilic addition cascade to form vinyl fluoride intermediates, which undergo rapid hydrofluorination to yield gem-difluorinated products, thereby transforming aliphatic CF3 groups in commercial building blocks and medicinal compounds into diverse α,α-difluoro(thio)ether motifs.

Transition-Metal Hydride Catalysis Meets Nitrenoid Transfer: Design Principles for Precision C–N Bond Formation
X. Lyu,† H. Choi† & S. Chang*
Acc. Chem. Res. 2026, ASAP (DOI: 10.1021/acs.accounts.6c00040) 🔓
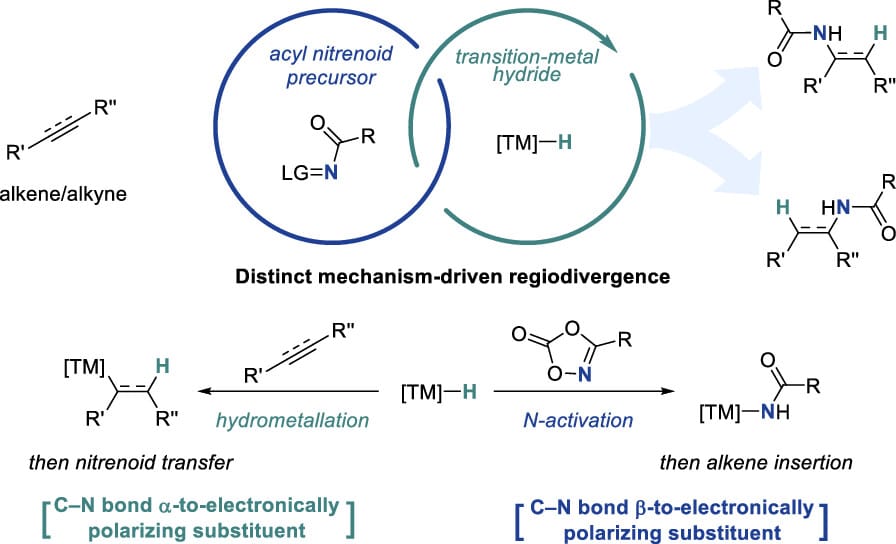
Transition-metal hydride (TMH) catalysis is a powerful approach for hydroamination, enabling C–N bond formation from alkenes and alkynes under mild conditions. Conventional TMH pathways involve hydrometalation, which directs regioselectivity to electronically activated sites. This Account describes the combination of TMH catalysis with nitrenoid transfer, revealing two mechanistically distinct hydroamidation regimes dictated by step order. Using dioxazolones as nitrenoid precursors, a canonical pathway was established in which hydrometalation precedes nitrenoid transfer, enabling predictable regioselective hydroamidation. In contrast, NiH catalysis can initiate by nitrenoid activation, forming a Ni-amido intermediate that engages the alkene, shifting selectivity control to C–N bond formation and enabling otherwise inaccessible regioselectivity.

From Crude to Crisis
🛢️ From Crude to Crisis. The bulk chemical industry is now starting to feel the effects of the war in Iran as the near-shutdown of the Strait of Hormuz—that place we all definitely knew about a month ago—halts a critical supply route. Up to 80% of Asia’s naphtha, a crude oil–derived feedstock used to make basic chemicals, comes from the Middle East. With shipments stalled, petrochemical firms are suspending contracts, cutting operating rates and delaying plant restarts.
The risk is particularly challenging for China, which imports ~12 million barrels of crude oil per day, about 14% of which comes from Iran. Naphtha is steam-cracked to produce ethylene, a crucial building block that underpins plastics, solvents and countless everyday materials. As supply tightens, naphtha prices have already surged past $1,000 per tonne. The disruption goes beyond petrochemicals with strikes on Qatar shutting key facilities and removing roughly one-third of global helium supply. The country also accounts for around 14% of global urea capacity (a key nitrogen fertiliser for food production) and ~20% of liquid natural gas supply.
With 20% of global oil flows passing through the Strait of Hormuz, the industry now faces both feedstock shortages and rising energy costs. Disruptions to bulk chemical production can quickly cascade into everyday research laboratories. For example, during the 2008 Beijing Olympics, factory shutdowns near and upwind of Beijing (implemented to improve air quality) reduced acrylonitrile production, inadvertently cutting acetonitrile supply, which is formed as a by-product. Since acetonitrile is so widely used in HPLC and LCMS workflows, laboratories worldwide suddenly faced severe shortages. If disruption persists in the Strait, it could reshape global supply chains, accelerating a shift away from Middle Eastern feedstocks.
That’s all for this issue! Have a great week and we’ll see you next Monday.

Reply